Metabolically active neutrophils represent a permissive niche for Mycobacterium tuberculosis
- PMID: 38844208
- PMCID: PMC11493682
- DOI: 10.1016/j.mucimm.2024.05.007
Metabolically active neutrophils represent a permissive niche for Mycobacterium tuberculosis
Abstract
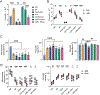
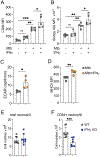
Mycobacterium tuberculosis (Mtb)-infected neutrophils are often found in the airways of patients with active tuberculosis (TB), and excessive recruitment of neutrophils to the lung is linked to increased bacterial burden and aggravated pathology in TB. The basis for the permissiveness of neutrophils for Mtb and the ability to be pathogenic in TB has been elusive. Here, we identified metabolic and functional features of neutrophils that contribute to their permissiveness in Mtb infection. Using single-cell metabolic and transcriptional analyses, we found that neutrophils in the Mtb-infected lung displayed elevated mitochondrial metabolism, which was largely attributed to the induction of activated neutrophils with enhanced metabolic activities. The activated neutrophil subpopulation was also identified in the lung granulomas from Mtb-infected non-human primates. Functionally, activated neutrophils harbored more viable bacteria and displayed enhanced lipid uptake and accumulation. Surprisingly, we found that interferon-γ promoted the activation of lung neutrophils during Mtb infection. Lastly, perturbation of lipid uptake pathways selectively compromised Mtb survival in activated neutrophils. These findings suggest that neutrophil heterogeneity and metabolic diversity are key to their permissiveness for Mtb and that metabolic pathways in neutrophils represent potential host-directed therapeutics in TB.
Copyright © 2024 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
DECLARATIONS OF COMPETING INTEREST
The authors have no competing interests to declare.
Figures





Similar articles
-
Fatty acid metabolism in neutrophils promotes lung damage and bacterial replication during tuberculosis.PLoS Pathog. 2024 Oct 4;20(10):e1012188. doi: 10.1371/journal.ppat.1012188. eCollection 2024 Oct. PLoS Pathog. 2024. PMID: 39365825 Free PMC article.
-
Permissive lung neutrophils facilitate tuberculosis immunopathogenesis in male phagocyte NADPH oxidase-deficient mice.PLoS Pathog. 2024 Aug 23;20(8):e1012500. doi: 10.1371/journal.ppat.1012500. eCollection 2024 Aug. PLoS Pathog. 2024. PMID: 39178329 Free PMC article.
-
Neutrophils in Tuberculosis-Associated Inflammation and Lung Pathology.Front Immunol. 2020 May 27;11:962. doi: 10.3389/fimmu.2020.00962. eCollection 2020. Front Immunol. 2020. PMID: 32536917 Free PMC article. Review.
-
Neutrophils express pro- and anti-inflammatory cytokines in granulomas from Mycobacterium tuberculosis-infected cynomolgus macaques.Mucosal Immunol. 2019 Nov;12(6):1370-1381. doi: 10.1038/s41385-019-0195-8. Epub 2019 Aug 21. Mucosal Immunol. 2019. PMID: 31434990 Free PMC article.
-
Neutrophils and Close Relatives in the Hypoxic Environment of the Tuberculous Granuloma: New Avenues for Host-Directed Therapies?Front Immunol. 2019 Mar 12;10:417. doi: 10.3389/fimmu.2019.00417. eCollection 2019. Front Immunol. 2019. PMID: 30915076 Free PMC article. Review.
Cited by
-
The role of neutrophil response in lung damage and post-tuberculosis lung disease: a translational narrative review.Front Immunol. 2025 Mar 7;16:1528074. doi: 10.3389/fimmu.2025.1528074. eCollection 2025. Front Immunol. 2025. PMID: 40124364 Free PMC article. Review.
-
Bioinformatics-Driven Identification of Ferroptosis-Related Gene Signatures Distinguishing Active and Latent Tuberculosis.Genes (Basel). 2025 Jun 18;16(6):716. doi: 10.3390/genes16060716. Genes (Basel). 2025. PMID: 40565608 Free PMC article.
-
Fatty acid metabolism in neutrophils promotes lung damage and bacterial replication during tuberculosis.PLoS Pathog. 2024 Oct 4;20(10):e1012188. doi: 10.1371/journal.ppat.1012188. eCollection 2024 Oct. PLoS Pathog. 2024. PMID: 39365825 Free PMC article.
-
The immunometabolic topography of tuberculosis granulomas governs cellular organization and bacterial control.bioRxiv [Preprint]. 2025 Feb 23:2025.02.18.638923. doi: 10.1101/2025.02.18.638923. bioRxiv. 2025. PMID: 40027668 Free PMC article. Preprint.
-
Multi-Omics Integration Reveals Mitochondrial Gene Regulation as a Determinant of Tuberculosis Susceptibility: A Mendelian Randomization Approach.Biomedicines. 2025 Mar 19;13(3):749. doi: 10.3390/biomedicines13030749. Biomedicines. 2025. PMID: 40149725 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

