The tumour microenvironment, treatment resistance and recurrence in glioblastoma
- PMID: 38844944
- PMCID: PMC11155041
- DOI: 10.1186/s12967-024-05301-9
The tumour microenvironment, treatment resistance and recurrence in glioblastoma
Abstract
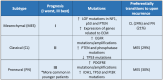
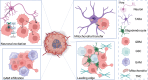
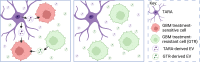
The adaptability of glioblastoma (GBM) cells, encouraged by complex interactions with the tumour microenvironment (TME), currently renders GBM an incurable cancer. Despite intensive research, with many clinical trials, GBM patients rely on standard treatments including surgery followed by radiation and chemotherapy, which have been observed to induce a more aggressive phenotype in recurrent tumours. This failure to improve treatments is undoubtedly a result of insufficient models which fail to incorporate components of the human brain TME. Research has increasingly uncovered mechanisms of tumour-TME interactions that correlate to worsened patient prognoses, including tumour-associated astrocyte mitochondrial transfer, neuronal circuit remodelling and immunosuppression. This tumour hijacked TME is highly implicated in driving therapy resistance, with further alterations within the TME and tumour resulting from therapy exposure inducing increased tumour growth and invasion. Recent developments improving organoid models, including aspects of the TME, are paving an exciting future for the research and drug development for GBM, with the hopes of improving patient survival growing closer. This review focuses on GBMs interactions with the TME and their effect on tumour pathology and treatment efficiency, with a look at challenges GBM models face in sufficiently recapitulating this complex and highly adaptive cancer.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Joseph JV, Blaavand MS, Daubon T, Kruyt FAE, Thomsen MK. Three-dimensional culture models to study glioblastoma—current trends and future perspectives. Curr Opin Pharmacol. 2021;61:91–97. - PubMed
-
- Miller DM, et al. Untangling the web of glioblastoma treatment resistance using a multi-omic and multidisciplinary approach. Am J Med Sci. 2023;366:185–198. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

