Spinal cord neurone loss and foot placement changes in a rat knock-in model of amyotrophic lateral sclerosis Type 8
- PMID: 38846532
- PMCID: PMC11154649
- DOI: 10.1093/braincomms/fcae184
Spinal cord neurone loss and foot placement changes in a rat knock-in model of amyotrophic lateral sclerosis Type 8
Abstract
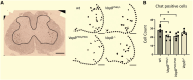
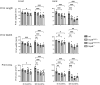
Amyotrophic lateral sclerosis is an age-dependent cell type-selective degenerative disease. Genetic studies indicate that amyotrophic lateral sclerosis is part of a spectrum of disorders, ranging from spinal muscular atrophy to frontotemporal dementia that share common pathological mechanisms. Amyotrophic lateral sclerosis Type 8 is a familial disease caused by mis-sense mutations in VAPB. VAPB is localized to the cytoplasmic surface of the endoplasmic reticulum, where it serves as a docking point for cytoplasmic proteins and mediates inter-organelle interactions with the endoplasmic reticulum membrane. A gene knock-in model of amyotrophic lateral sclerosis Type 8 based on the VapBP56S mutation and VapB gene deletion has been generated in rats. These animals display a range of age-dependent phenotypes distinct from those previously reported in mouse models of amyotrophic lateral sclerosis Type 8. A loss of motor neurones in VapBP56S/+ and VapBP56S/P56S animals is indicated by a reduction in the number of large choline acetyl transferase-staining cells in the spinal cord. VapB-/- animals exhibit a relative increase in cytoplasmic TDP-43 levels compared with the nucleus, but no large protein aggregates. Concomitant with these spinal cord pathologies VapBP56S/+ , VapBP56S/P56S and VapB-/- animals exhibit age-dependent changes in paw placement and exerted pressures when traversing a CatWalk apparatus, consistent with a somatosensory dysfunction. Extramotor dysfunction is reported in half the cases of motor neurone disease, and this is the first indication of an associated sensory dysfunction in a rodent model of amyotrophic lateral sclerosis. Different rodent models may offer complementary experimental platforms with which to understand the human disease.
Keywords: ALS8; VAPB; motor neurone disease.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Guarantors of Brain.
Conflict of interest statement
P.A. Skehel is an Associate Editor at Brain Communications.
Figures



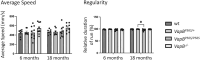

Similar articles
-
Amyotrophic lateral sclerosis-related VAPB P56S mutation differentially affects the function and survival of corticospinal and spinal motor neurons.Hum Mol Genet. 2013 Nov 1;22(21):4293-305. doi: 10.1093/hmg/ddt279. Epub 2013 Jun 13. Hum Mol Genet. 2013. PMID: 23771029 Free PMC article.
-
Vapb/Amyotrophic lateral sclerosis 8 knock-in mice display slowly progressive motor behavior defects accompanying ER stress and autophagic response.Hum Mol Genet. 2015 Nov 15;24(22):6515-29. doi: 10.1093/hmg/ddv360. Epub 2015 Sep 11. Hum Mol Genet. 2015. PMID: 26362257 Free PMC article.
-
Pathomechanisms of ALS8: altered autophagy and defective RNA binding protein (RBP) homeostasis due to the VAPB P56S mutation.Cell Death Dis. 2021 May 10;12(5):466. doi: 10.1038/s41419-021-03710-y. Cell Death Dis. 2021. PMID: 33972508 Free PMC article.
-
The Link between VAPB Loss of Function and Amyotrophic Lateral Sclerosis.Cells. 2021 Jul 23;10(8):1865. doi: 10.3390/cells10081865. Cells. 2021. PMID: 34440634 Free PMC article. Review.
-
Mutant VAPB: Culprit or Innocent Bystander of Amyotrophic Lateral Sclerosis?Contact (Thousand Oaks). 2021 Jun 15;4:25152564211022515. doi: 10.1177/25152564211022515. eCollection 2021 Jan-Dec. Contact (Thousand Oaks). 2021. PMID: 37366377 Free PMC article. Review.
Cited by
-
Automated gait analysis indicates efficacy of T-type calcium channel inhibition for mitigation of disrupted calcium signalling in an SCA5 mouse model.Sci Rep. 2025 Jul 1;15(1):20990. doi: 10.1038/s41598-025-05511-1. Sci Rep. 2025. PMID: 40594196 Free PMC article.
References
-
- Di L, Chen H, Da Y, Wang S, Shen XM. Atypical familial amyotrophic lateral sclerosis with initial symptoms of pain or tremor in a Chinese family harboring VAPB-P56S mutation. J Neurol. 2016;263(2):263–268. - PubMed
-
- Millecamps S, Salachas F, Cazeneuve C, et al. SOD1, ANG, VAPB, TARDBP, and FUS mutations in familial amyotrophic lateral sclerosis: Genotype-phenotype correlations. J Med Genet. 2010;47(8):554–560. - PubMed
LinkOut - more resources
Full Text Sources
