RASopathies - what they reveal about RAS/MAPK signaling in skeletal muscle development
- PMID: 38847227
- PMCID: PMC11179721
- DOI: 10.1242/dmm.050609
RASopathies - what they reveal about RAS/MAPK signaling in skeletal muscle development
Abstract
RASopathies are rare developmental genetic syndromes caused by germline pathogenic variants in genes that encode components of the RAS/mitogen-activated protein kinase (MAPK) signal transduction pathway. Although the incidence of each RASopathy syndrome is rare, collectively, they represent one of the largest groups of multiple congenital anomaly syndromes and have severe developmental consequences. Here, we review our understanding of how RAS/MAPK dysregulation in RASopathies impacts skeletal muscle development and the importance of RAS/MAPK pathway regulation for embryonic myogenesis. We also discuss the complex interactions of this pathway with other intracellular signaling pathways in the regulation of skeletal muscle development and growth, and the opportunities that RASopathy animal models provide for exploring the use of pathway inhibitors, typically used for cancer treatment, to correct the unique skeletal myopathy caused by the dysregulation of this pathway.
Keywords: Cardio-facio-cutaneous syndrome; Costello syndrome; Myopathy; Neurofibromatosis type 1; RAS pathway; RASopathy; Rare disorder; Skeletal myogenesis; Treatment.
© 2024. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures
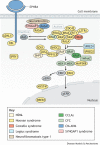
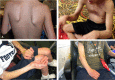
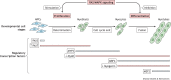
Similar articles
-
Ras/MAPK dysregulation in development causes a skeletal myopathy in an activating BrafL597V mouse model for cardio-facio-cutaneous syndrome.Dev Dyn. 2021 Aug;250(8):1074-1095. doi: 10.1002/dvdy.309. Epub 2021 Feb 13. Dev Dyn. 2021. PMID: 33522658
-
Skeletal muscle pathology in Costello and cardio-facio-cutaneous syndromes: developmental consequences of germline Ras/MAPK activation on myogenesis.Am J Med Genet C Semin Med Genet. 2011 May 15;157C(2):104-14. doi: 10.1002/ajmg.c.30298. Epub 2011 Apr 14. Am J Med Genet C Semin Med Genet. 2011. PMID: 21495178
-
The RASopathies.Annu Rev Genomics Hum Genet. 2013;14:355-69. doi: 10.1146/annurev-genom-091212-153523. Epub 2013 Jul 15. Annu Rev Genomics Hum Genet. 2013. PMID: 23875798 Free PMC article. Review.
-
Noonan, Costello and cardio-facio-cutaneous syndromes: dysregulation of the Ras-MAPK pathway.Expert Rev Mol Med. 2008 Dec 9;10:e37. doi: 10.1017/S1462399408000902. Expert Rev Mol Med. 2008. PMID: 19063751 Review.
-
Autism traits in the RASopathies.J Med Genet. 2014 Jan;51(1):10-20. doi: 10.1136/jmedgenet-2013-101951. Epub 2013 Oct 7. J Med Genet. 2014. PMID: 24101678 Free PMC article.
Cited by
-
Translating multiscale research in rare disease.Dis Model Mech. 2024 Jun 1;17(6):dmm052009. doi: 10.1242/dmm.052009. Epub 2024 Jul 10. Dis Model Mech. 2024. PMID: 38982973 Free PMC article.
-
Investigating the Impact of Circulating MicroRNAs on Knee and Hip Osteoarthritis: Causal Links, Biological Mechanisms, and Drug Interactions.Int J Mol Sci. 2024 Dec 31;26(1):283. doi: 10.3390/ijms26010283. Int J Mol Sci. 2024. PMID: 39796139 Free PMC article.
-
A 10-Year Review on Advancements in Identifying and Treating Intellectual Disability Caused by Genetic Variations.Genes (Basel). 2024 Aug 24;15(9):1118. doi: 10.3390/genes15091118. Genes (Basel). 2024. PMID: 39336708 Free PMC article. Review.
-
Advancing edge-based clustering and graph embedding for biological network analysis: a case study in RASopathies.Brief Bioinform. 2025 Jul 2;26(4):bbaf320. doi: 10.1093/bib/bbaf320. Brief Bioinform. 2025. PMID: 40619812 Free PMC article.
-
Intensive Value Utilization of Food-Derived Marine Immunoactive Peptides: Optimizing the Process Yield and Improving the Delivery Efficiency According to the Immune Activity Mechanism and Assisted Enzymatic Hydrolysis.Food Sci Nutr. 2025 Aug 5;13(8):e70578. doi: 10.1002/fsn3.70578. eCollection 2025 Aug. Food Sci Nutr. 2025. PMID: 40766787 Free PMC article. Review.
References
-
- Alessandrini, A., Brott, B. K. and Erikson, R. L. (1997). Differential expression of MEK1 and MEK2 during mouse development. Cell Growth Differ. 8, 505-511. - PubMed
-
- Amyere, M., Revencu, N., Helaers, R., Pairet, E., Baselga, E., Cordisco, M., Chung, W., Dubois, J., Lacour, J. P., Martorell, L.et al. (2017). Germline loss-of-function mutations in EPHB4 cause a second form of capillary malformation-arteriovenous malformation (CM-AVM2) deregulating RAS-MAPK signaling. Circulation 136, 1037-1048. 10.1161/CIRCULATIONAHA.116.026886 - DOI - PubMed
-
- Andreadi, C., Cheung, L. K., Giblett, S., Patel, B., Jin, H., Mercer, K., Kamata, T., Lee, P., Williams, A., McMahon, M.et al. (2012). The intermediate-activity (L597V)BRAF mutant acts as an epistatic modifier of oncogenic RAS by enhancing signaling through the RAF/MEK/ERK pathway. Genes Dev. 26, 1945-1958. 10.1101/gad.193458.112 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

