International Map of Axial Spondyloarthritis (IMAS): results from the perspective of 5557 patients from 27 countries around the globe
- PMID: 38851236
- PMCID: PMC11163687
- DOI: 10.1136/rmdopen-2023-003504
International Map of Axial Spondyloarthritis (IMAS): results from the perspective of 5557 patients from 27 countries around the globe
Abstract
Background: The International Map of Axial Spondyloarthritis (IMAS) is a global initiative aimed to assess the impact and burden of axial spondyloarthritis (axSpA) and identify the unmet needs from the patient's perspective.
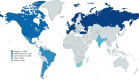
Method: IMAS is a collaboration between the Axial Spondyloarthritis International Federation (ASIF), the University of Seville, Novartis Pharma AG and steered by a scientific committee. IMAS collected information through an online cross-sectional survey (2017-2022) from unselected patients with axSpA from Europe, Asia, North America, Latin America and Africa who completed a comprehensive questionnaire containing over 120 items.
Results: 5557 patients with axSpA participated in IMAS. Mean age was 43.9 ±12.8 years, 55.4% were female, 46.2% had a university education and 51.0% were employed. The mean diagnostic delay was 7.4 ±9.0 years (median: 4.0), and the mean symptom duration was 17.1 ±13.3 years. 75.0% of patients had active disease (Bath Ankylosing Spondylitis Disease Activity Index ≥4), and 59.4% reported poor mental health (12-item General Health Questionnaire ≥3). In the year before the survey, patients had visited primary care physicians 4.6 times and the rheumatologist 3.6 times. 78.6% had taken non-steroidal anti-inflammatory drug ever, 48.8% biological disease-modifying antirheumatic drugs and 43.6% conventional synthetic disease-modifying antirheumatic drugs. Patients's greatest fear was disease progression (55.9%), while the greatest hope was to be able to relieve pain (54.2%).
Conclusions: IMAS shows the global profile of patients with axSpA, highlighting unmet needs, lengthy delays in diagnosis and high burden of disease in patients with axSpA worldwide. This global information will enable more detailed investigations to obtain evidence on the critical issues that matter to patients around the world to improve their care and quality of life.
Keywords: Patient Reported Outcome Measures; Patient perspective; Spondylitis, Ankylosing.
© Author(s) (or their employer(s)) 2024. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: Marco Garrido-Cumbrera grant/research support from: Novartis, Denis Poddubnyy Speakers bureau: AbbVie, BMS, Celgene, Janssen, Lilly, MSD, Novartis, Pfizer, Roche and UCB, 13 grant/research support from: AbbVie, MSD, Novartis, and Pfizer, Fernando Sommerfleck Speakers bureau: Abbvie, Eli Lilly, Janssen, Novartis, consultant of: Abbvie, Novartis, Janssen, Christine Bundy Speakers bureau: AbbVie, Celgene, Janssen, Lilly, Novartis and Pfizer, Souzi Makri Consultant of: Novartis, GSK and Bayer, José Correa-Fernández: none declared, Shashank Akerkar Speakers bureau: Pfizer, Novartis, Eli Lilly, Jansen, Jo Lowe Grant/research support from: no personal funding, but ASIF has received funding from Novartis, UCB, Lilly, Abbvie, Boehringer Ingleheim, Pfizer, Janssen, Elie Karam: none declared, Laura Christen Employee of: Novartis employment and stock ownership, Victoria Navarro-Compán Speakers bureau: AbbVie, Eli Lilly, Janssen, MSD, Novartis, Pfizer, UCB Pharma, consultant of: AbbVie, Eli Lilly, Galapagos, MoonLake, MSD, Novartis, Pfizer, UCB Pharma, grant/research support from: AbbVie, Novartis.
Figures
References
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials