Sub-100-fs energy transfer in coenzyme NADH is a coherent process assisted by a charge-transfer state
- PMID: 38851775
- PMCID: PMC11162464
- DOI: 10.1038/s41467-024-48871-4
Sub-100-fs energy transfer in coenzyme NADH is a coherent process assisted by a charge-transfer state
Erratum in
-
Author Correction: Sub-100-fs energy transfer in coenzyme NADH is a coherent process assisted by a charge-transfer state.Nat Commun. 2024 Sep 10;15(1):7912. doi: 10.1038/s41467-024-52280-y. Nat Commun. 2024. PMID: 39256436 Free PMC article. No abstract available.
Abstract
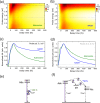
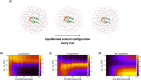
Excitation energy transfer (EET) is a key photoinduced process in biological chromophoric assemblies. Here we investigate the factors which can drive EET into efficient ultrafast sub-ps regimes. We demonstrate how a coherent transport of electronic population could facilitate this in water solvated NADH coenzyme and uncover the role of an intermediate dark charge-transfer state. High temporal resolution ultrafast optical spectroscopy gives a 54±11 fs time constant for the EET process. Nonadiabatic quantum dynamical simulations computed through the time-evolution of multidimensional wavepackets suggest that the population transfer is mediated by photoexcited molecular vibrations due to strong coupling between the electronic states. The polar aqueous solvent environment leads to the active participation of a dark charge transfer state, accelerating the vibronically coherent EET process in favorably stacked conformers and solvent cavities. Our work demonstrates how the interplay of structural and environmental factors leads to diverse pathways for the EET process in flexible heterodimers and provides general insights relevant for coherent EET processes in stacked multichromophoric aggregates like DNA strands.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Dynamics and quantumness of excitation energy transfer through a complex quantum network.Phys Rev E Stat Nonlin Soft Matter Phys. 2014 Oct;90(4):042140. doi: 10.1103/PhysRevE.90.042140. Epub 2014 Oct 27. Phys Rev E Stat Nonlin Soft Matter Phys. 2014. PMID: 25375471
-
Ultrafast Fluorescence Signals from β-Dihydronicotinamide Adenine Dinucleotide: Resonant Energy Transfer in the Folded and Unfolded Forms.J Phys Chem B. 2020 Jan 23;124(3):519-530. doi: 10.1021/acs.jpcb.9b10012. Epub 2020 Jan 8. J Phys Chem B. 2020. PMID: 31876417
-
Role of Pigment-Protein Coupling in the Energy Transport Dynamics in the Fenna-Matthews-Olson Complex.J Phys Chem B. 2021 Nov 4;125(43):11884-11892. doi: 10.1021/acs.jpcb.1c06844. Epub 2021 Oct 20. J Phys Chem B. 2021. PMID: 34669415
-
Intramolecular Vibrations in Excitation Energy Transfer: Insights from Real-Time Path Integral Calculations.Annu Rev Phys Chem. 2022 Apr 20;73:349-375. doi: 10.1146/annurev-physchem-090419-120202. Epub 2022 Jan 26. Annu Rev Phys Chem. 2022. PMID: 35081322 Review.
-
Review: Pathway analysis for peptide-mediated electronic coupling in the super-exchange mechanism of ET and EET.Biopolymers. 2013;100(1):100-13. doi: 10.1002/bip.22142. Biopolymers. 2013. PMID: 23335172 Review.
References
-
- Ashkenazi, G., Kosloff, R. & Ratner, M. A. Photoexcited electron transfer: short-time dynamics and turnover control by dephasing, relaxation, and mixing. J. Am. Chem. Soc.121, 3386–3395 (1999).10.1021/ja981998p - DOI
-
- Xu, D. & Schulten, K. Coupling of protein motion to electron transfer in a photosynthetic reaction center: investigating the low temperature behavior in the framework of the spin—boson model. Chem. Phys.182, 91–117 (1994).10.1016/0301-0104(94)00016-6 - DOI
-
- Ando, K. & Sumi, H. Nonequilibrium oscillatory electron transfer in bacterial photosynthesis. J. Phys. Chem. B102, 10991–11000 (1998).10.1021/jp982659l - DOI
-
- Olaya-Castro, A. & Scholes, G. D. Energy transfer from Förster–Dexter theory to quantum coherent light-harvesting. Int. Rev. Phys. Chem.30, 49–77 (2011).10.1080/0144235X.2010.537060 - DOI
-
- Simpson, W. T. & Peterson, D. L. Coupling strength for resonance force transfer of electronic energy in van der waals solids. J. Chem. Phys.26, 588–593 (1957).10.1063/1.1743351 - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

