Spinal cord injury-induced metabolic impairment and steatohepatitis develops in non-obese rats and is exacerbated by premorbid obesity
- PMID: 38852834
- PMCID: PMC11874686
- DOI: 10.1016/j.expneurol.2024.114847
Spinal cord injury-induced metabolic impairment and steatohepatitis develops in non-obese rats and is exacerbated by premorbid obesity
Abstract
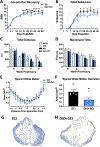
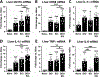
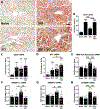
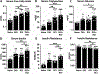
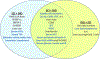
Impaired sensorimotor functions are prominent complications of spinal cord injury (SCI). A clinically important but less obvious consequence is development of metabolic syndrome (MetS), including increased adiposity, hyperglycemia/insulin resistance, and hyperlipidemia. MetS predisposes SCI individuals to earlier and more severe diabetes and cardiovascular disease compared to the general population, which trigger life-threatening complications (e.g., stroke, myocardial infarcts). Although each comorbidity is known to be a risk factor for diabetes and other health problems in obese individuals, their relative contribution or perceived importance in propagating systemic pathology after SCI has received less attention. This could be explained by an incomplete understanding of MetS promoted by SCI compared with that from the canonical trigger diet-induced obesity (DIO). Thus, here we compared metabolic-related outcomes after SCI in lean rats to those of uninjured rats with DIO. Surprisingly, SCI-induced MetS features were equal to or greater than those in obese uninjured rats, including insulin resistance, endotoxemia, hyperlipidemia, liver inflammation and steatosis. Considering the endemic nature of obesity, we also evaluated the effect of premorbid obesity in rats receiving SCI; the combination of DIO + SCI exacerbated MetS and liver pathology compared to either alone, suggesting that obese individuals that sustain a SCI are especially vulnerable to metabolic dysfunction. Notably, premorbid obesity also exacerbated intraspinal lesion pathology and worsened locomotor recovery after SCI. Overall, these results highlight that normal metabolic function requires intact spinal circuitry and that SCI is not just a sensory-motor disorder, but also has significant metabolic consequences.
Keywords: Cytokines; Diet-induced obesity; Insulin resistance; Kupffer cells; Locomotor recovery, alanine transferase; MASLD, metabolic dysfunction associated steatohepatitis (MASH); Metabolic syndrome; Non-alcoholic fatty liver disease; Non-alcoholic steatohepatitis (NASH).
Copyright © 2024 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare no conflicts of interest.
Figures
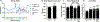



References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

