Efficacy and Safety of Compound Kushen Injection for Advanced Colorectal Cancer: A Systematic Review and Meta-Analysis of Randomized Clinical Trials with Trial Sequential Analysis
- PMID: 38853681
- PMCID: PMC11163932
- DOI: 10.1177/15347354241258458
Efficacy and Safety of Compound Kushen Injection for Advanced Colorectal Cancer: A Systematic Review and Meta-Analysis of Randomized Clinical Trials with Trial Sequential Analysis
Abstract
Backgrounds: Colorectal cancer (CRC) is one of the common malignant tumors, with a gradually increasing incidence. Due to late detection and poor sensitivity to chemotherapy, it has become a difficult problem in tumor prevention and treatment at present. Exploring or discovering new combinations is a significant strategy for the treatment of CRC. Compound kushen injection (CKI) is a traditional Chinese medicine injection extracted from Sophora flavescens Ait. and Smilax glabra Roxb., which is widely used in the comprehensive treatment of CRC in China. This systematic review is aimed to ascertain the clinical efficacy and safety of CKI combined with chemotherapy in the treatment of advanced CRC based on available data. On this basis, the specific application of CKI in combination with chemotherapy in clinical practice is further discussed.
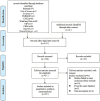
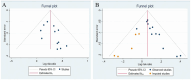
Methods: PubMed, Web of Science, the Cochrane Library, EMBASE, China National Knowledge Infrastructure, Chinese Scientific Journals Database, Wanfang Database, Chinese Biomedicine Database Searches, the Chinese Clinical Trial Registry, and ClinicalTrials.gov were searched systematically, from inception to April 20, 2024. We adopted the ROB2 tool to assess quality of the included trials, Stata 16 for data analysis, and evaluated the publication bias with the funnel plot and Egger's test. The quality of the evidence was justified according to GRADE. We also used trial sequential analysis (TSA) to calculate the final required sample size in this meta-analysis and to verify whether the results present a reliable conclusion. The protocol for this systematic review was registered on PROSPERO (CRD42022380106) and has been published.
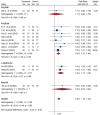
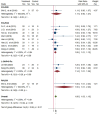
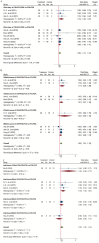
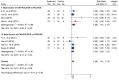
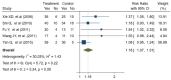
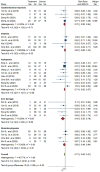
Results: Sixteen trials that examined 1378 patients were included in this study. Meta-analysis revealed that compared with chemotherapy, objective response rate (ORR, RR = 1.30, 95% CI: 1.18-1.44), disease control rate (DCR, RR = 1.08, 95% CI: 1.03-1.13), and KPS score improvement rate were improved (RR = 1.18, 95% CI: 1.07-1.31) by the combination of CKI and chemotherapy in patients with advanced CRC. Additionally, CKI combined with chemotherapy was associated with lower adverse reactions such as leukopenia (RR = 0.74, 95% CI: 0.62-0.87), thrombocytopenia (RR = 0.68, 95% CI: 0.49-0.94), gastrointestinal reactions (RR = 0.72, 95% CI: 0.55-0.94), and liver damage (RR = 0.48, 95% CI: 0.30-0.79), higher CD4+ ratio (MD = 9.70, 95% CI:8.73-10.68) and CD4+/CD8+ ratio (MD = 0.25, 95% CI: 0.22-0.28), and lower CD8+ T cell ratio (MD = -5.25, 95% CI: -5.94 to -4.56). Subgroup analysis demonstrated that ORR and DCR in patients with advanced CRC were improved when CKI combined with FOLFOX and 5Fu + L-OHP. Both 15 and 20 ml/day of CKI combined with FOLFOX provided a significant effect in ORR. Moreover, ORR was improved when the accumulated CKI dose reached 280 ml per course and 420 ml in total. 7 days/course as well as 14 days/course of CKI combined with FOLFOX were effective durations in ORR. As for DCR, 7 days/course of CKI combined with FOLFOX could improve efficacy. Furthermore, CKI + FOLFOX may be useful in ORR and DCR for at least 4 cycles of combination therapies. The TSA showed that firm results in ORR and DCR were established and additional trials were unlikely to change the results.
Conclusion: CKI combined with chemotherapy provides a statistically significant and clinically important effect in the improvement of ORR, DCR, performance status, ADR reduction, and immune function in patients with CRC. However, more rigorously designed, large-scale, and multi-center RCTs are needed in the future.
Keywords: Compound Kushen injection; TSA; chemotherapeutic drugs; colorectal cancer; traditional Chinese medicine.
Conflict of interest statement
Declaration of Conflicting InterestsThe author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures









Similar articles
-
Effect of compound kushen injection on immune function in patients with colorectal cancer: a systematic review and meta-analysis.Front Pharmacol. 2025 Apr 3;16:1565031. doi: 10.3389/fphar.2025.1565031. eCollection 2025. Front Pharmacol. 2025. PMID: 40248100 Free PMC article.
-
Efficacy and safety of compound kushen injection for treating advanced colorectal cancer: A protocol for a systematic review and meta-analysis.Heliyon. 2024 Feb 28;10(5):e26981. doi: 10.1016/j.heliyon.2024.e26981. eCollection 2024 Mar 15. Heliyon. 2024. PMID: 38463847 Free PMC article.
-
Clinical efficacy and safety of NSCLC ancillary treatment with compound Kushen injection through immunocompetence regulation: A systematic review and meta-analysis.Phytomedicine. 2022 Sep;104:154315. doi: 10.1016/j.phymed.2022.154315. Epub 2022 Jul 6. Phytomedicine. 2022. PMID: 35868145
-
Clinical Effectiveness and Safety of Chinese Herbal Medicine Compound Kushen Injection as an Add-On Treatment for Breast Cancer: A Systematic Review and Meta-Analysis.Evid Based Complement Alternat Med. 2022 Jan 10;2022:8118408. doi: 10.1155/2022/8118408. eCollection 2022. Evid Based Complement Alternat Med. 2022. PMID: 35047051 Free PMC article.
-
Combination of Compound Kushen injection with first-line treatment versus first-line treatment alone for advanced colorectal cancer: a study protocol for a multicenter, openlabel, randomized controlled trial.BMC Complement Med Ther. 2024 Dec 31;24(1):429. doi: 10.1186/s12906-024-04725-6. BMC Complement Med Ther. 2024. PMID: 39741233 Free PMC article.
Cited by
-
Effect of compound kushen injection on immune function in patients with colorectal cancer: a systematic review and meta-analysis.Front Pharmacol. 2025 Apr 3;16:1565031. doi: 10.3389/fphar.2025.1565031. eCollection 2025. Front Pharmacol. 2025. PMID: 40248100 Free PMC article.
-
Therapeutic potential and mechanistic insights of silibinin targeting cancer-associated fibroblasts in colorectal cancer.Front Pharmacol. 2025 Apr 2;16:1527871. doi: 10.3389/fphar.2025.1527871. eCollection 2025. Front Pharmacol. 2025. PMID: 40242441 Free PMC article.
References
-
- Sung H, Ferlay J, Siegel RL, et al.. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209-249. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

