This is a preprint.
Directly Measuring Atherogenic Lipoprotein Kinetics in Zebrafish with the Photoconvertible LipoTimer Reporter
- PMID: 38853962
- PMCID: PMC11160697
- DOI: 10.1101/2024.05.29.596423
Directly Measuring Atherogenic Lipoprotein Kinetics in Zebrafish with the Photoconvertible LipoTimer Reporter
Update in
-
Directly Measuring Atherogenic Lipoprotein Kinetics in Zebrafish With the Photoconvertible LipoTimer Reporter.Arterioscler Thromb Vasc Biol. 2025 Aug 21. doi: 10.1161/ATVBAHA.125.322969. Online ahead of print. Arterioscler Thromb Vasc Biol. 2025. PMID: 40836916
Abstract
Lipoprotein kinetics are a crucial factor in understanding lipoprotein metabolism since a prolonged time in circulation can contribute to the atherogenic character of apolipoprotein-B (ApoB)-containing lipoproteins (B-lps). Here, we report a method to directly measure lipoprotein kinetics in live developing animals. We developed a zebrafish geneticly encoded reporter, LipoTimer, in which endogenous ApoBb.1 is fused to the photoconvertible fluorophore Dendra2 which shift its emission profile from green to red upon UV exposure. By quantifying the red population of ApoB-Dendra2 over time, we found that B-lp turnover in wild-type larvae becomes faster as development proceeds. Mutants with impaired B-lp uptake or lipolysis present with increased B-lp levels and half-life. In contrast, mutants with impaired B-lp triglyceride loading display slightly fewer and smaller-B-lps, which have a significantly shorter B-lp half-life. Further, we showed that chronic high-cholesterol feeding is associated with a longer B-lp half-life in wild-type juveniles but does not lead to changes in B-lp half-life in lipolysis deficient apoC2 mutants. These data support the hypothesis that B-lp lipolysis is suppressed by the flood of intestinal-derived B-lps that follow a high-fat meal.
Keywords: apolipoprotein B; half-life; kinetics; lipoproteins; zebrafish.
Conflict of interest statement
Declaration of interests The authors have declared that no conflict of interest exists.
Figures
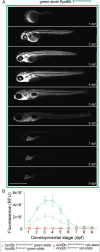
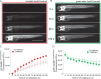

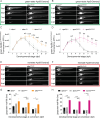
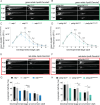

Similar articles
-
Directly Measuring Atherogenic Lipoprotein Kinetics in Zebrafish With the Photoconvertible LipoTimer Reporter.Arterioscler Thromb Vasc Biol. 2025 Aug 21. doi: 10.1161/ATVBAHA.125.322969. Online ahead of print. Arterioscler Thromb Vasc Biol. 2025. PMID: 40836916
-
Can a Liquid Biopsy Detect Circulating Tumor DNA With Low-passage Whole-genome Sequencing in Patients With a Sarcoma? A Pilot Evaluation.Clin Orthop Relat Res. 2025 Jan 1;483(1):39-48. doi: 10.1097/CORR.0000000000003161. Epub 2024 Jun 21. Clin Orthop Relat Res. 2025. PMID: 38905450
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
The Effect of Endocrine Disorders on Lipids and Lipoproteins.2023 Apr 6. In: Feingold KR, Ahmed SF, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, Muzumdar R, Purnell J, Rey R, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000–. 2023 Apr 6. In: Feingold KR, Ahmed SF, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, Muzumdar R, Purnell J, Rey R, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000–. PMID: 28121116 Free Books & Documents. Review.
References
-
- Feingold KR, Grunfeld C. Introduction to Lipids and Lipoproteins. In: Feingold KR, Anawalt B, Boyce A, Chrousos G, Dungan K, Grossman A, et al. , editors. Endotext. South Dartmouth (MA)2000.
-
- Schumaker VN, Phillips ML, Chatterton JE. Apolipoprotein B and low-density lipoprotein structure: implications for biosynthesis of triglyceride-rich lipoproteins. Adv Protein Chem. 1994;45:205–48. - PubMed
-
- Hussain MM, Kancha RK, Zhou Z, Luchoomun J, Zu H, Bakillah A. Chylomicron assembly and catabolism: role of apolipoproteins and receptors. Biochim Biophys Acta. 1996;1300(3):151–70. - PubMed
-
- Howard BV, Robbins DC, Sievers ML, Lee ET, Rhoades D, Devereux RB, et al. LDL cholesterol as a strong predictor of coronary heart disease in diabetic individuals with insulin resistance and low LDL: The Strong Heart Study. Arteriosclerosis, thrombosis, and vascular biology. 2000;20(3):830–5. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
