Protective effect of zinc gluconate on intestinal mucosal barrier injury in antibiotics and LPS-induced mice
- PMID: 38855764
- PMCID: PMC11157515
- DOI: 10.3389/fmicb.2024.1407091
Protective effect of zinc gluconate on intestinal mucosal barrier injury in antibiotics and LPS-induced mice
Abstract
Objective: The aim of the study is to investigate the function and mechanism of Zinc Gluconate (ZG) on intestinal mucosal barrier damage in antibiotics and Lipopolysaccharide (LPS)-induced mice.
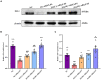
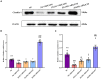
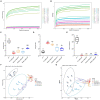
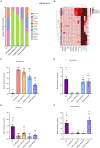
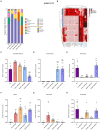
Methods: We established a composite mouse model by inducing intestinal mucosal barrier damage using antibiotics and LPS. The animals were divided into five groups: Control (normal and model) and experimental (low, medium, and high-dose ZG treatments). We evaluated the intestinal mucosal barrier using various methods, including monitoring body weight and fecal changes, assessing pathological damage and ultrastructure of the mouse ileum, analyzing expression levels of tight junction (TJ)-related proteins and genes, confirming the TLR4/NF-κB signaling pathway, and examining the structure of the intestinal flora.
Results: In mice, the dual induction of antibiotics and LPS led to weight loss, fecal abnormalities, disruption of ileocecal mucosal structure, increased intestinal barrier permeability, and disorganization of the microbiota structure. ZG restored body weight, alleviated diarrheal symptoms and pathological damage, and maintained the structural integrity of intestinal epithelial cells (IECs). Additionally, ZG reduced intestinal mucosal permeability by upregulating TJ-associated proteins (ZO-1, Occludin, Claudin-1, and JAM-A) and downregulating MLCK, thereby repairing intestinal mucosal barrier damage induced by dual induction of antibiotics and LPS. Moreover, ZG suppressed the TLR4/NF-κB signaling pathway, demonstrating anti-inflammatory properties and preserving barrier integrity. Furthermore, ZG restored gut microbiota diversity and richness, evidenced by increased Shannon and Observed features indices, and decreased Simpson's index. ZG also modulated the relative abundance of beneficial human gut bacteria (Bacteroidetes, Firmicutes, Verrucomicrobia, Parabacteroides, Lactobacillus, and Akkermansia) and harmful bacteria (Proteobacteria and Enterobacter), repairing the damage induced by dual administration of antibiotics and LPS.
Conclusion: ZG attenuates the dual induction of antibiotics and LPS-induced intestinal barrier damage and also protects the intestinal barrier function in mice.
Keywords: 16S rRNA; LPS; TLR4/NF-κB; antibiotics; gut microbiome; intestinal mucosal barrier; tight junction; zinc gluconate.
Copyright © 2024 Wang, Xiao, Wei, Su, Yang, Su, Lan, Chen, Huang and Shan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures







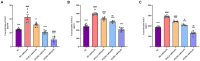

Similar articles
-
Ganluyin ameliorates DSS-induced ulcerative colitis by inhibiting the enteric-origin LPS/TLR4/NF-κB pathway.J Ethnopharmacol. 2022 May 10;289:115001. doi: 10.1016/j.jep.2022.115001. Epub 2022 Jan 24. J Ethnopharmacol. 2022. PMID: 35085745
-
Isoquercitrin alleviates lipopolysaccharide-induced intestinal mucosal barrier damage in mice by regulating TLR4/MyD88/NF-κB signaling pathway and intestinal flora.Food Funct. 2024 Jan 2;15(1):295-309. doi: 10.1039/d3fo03319h. Food Funct. 2024. PMID: 38084034
-
Jinzhi protects lipopolysaccharide-treated mice against mortality by repairing intestinal mucosal barrier damage and intestinal microecology.Biomed Pharmacother. 2020 Mar;123:109749. doi: 10.1016/j.biopha.2019.109749. Epub 2019 Dec 14. Biomed Pharmacother. 2020. PMID: 31846840
-
Gut Barrier Dysfunction and Microbiota Variations in Cryptosporidiosis: A Comprehensive Review.Vet Sci. 2025 Jan 23;12(2):85. doi: 10.3390/vetsci12020085. Vet Sci. 2025. PMID: 40005845 Free PMC article. Review.
-
Intestinal barrier permeability: the influence of gut microbiota, nutrition, and exercise.Front Physiol. 2024 Jul 8;15:1380713. doi: 10.3389/fphys.2024.1380713. eCollection 2024. Front Physiol. 2024. PMID: 39040079 Free PMC article. Review.
Cited by
-
Effect of maternal diet on gut bacteria and autism spectrum disorder in offspring.Front Cell Neurosci. 2025 Aug 6;19:1623576. doi: 10.3389/fncel.2025.1623576. eCollection 2025. Front Cell Neurosci. 2025. PMID: 40842563 Free PMC article. Review.
-
Dietary astragalin confers protection against lipopolysaccharide-induced intestinal mucosal barrier damage through mitigating inflammation and modulating intestinal microbiota.Front Nutr. 2024 Oct 2;11:1481203. doi: 10.3389/fnut.2024.1481203. eCollection 2024. Front Nutr. 2024. PMID: 39421621 Free PMC article.
-
Apolipoprotein A-I: Potential Protection Against Intestinal Injury Induced by Dietary Lipid.J Inflamm Res. 2024 Aug 28;17:5711-5721. doi: 10.2147/JIR.S468842. eCollection 2024. J Inflamm Res. 2024. PMID: 39219814 Free PMC article. Review.
-
Quinazolinone Derivative MR2938 Protects DSS-Induced Barrier Dysfunction in Mice Through Regulating Gut Microbiota.Pharmaceuticals (Basel). 2025 Jan 17;18(1):123. doi: 10.3390/ph18010123. Pharmaceuticals (Basel). 2025. PMID: 39861184 Free PMC article.
-
Elucidation of the mechanism of berberine against gastric mucosa injury in a rat model with chronic atrophic gastritis based on a combined strategy of multi-omics and molecular biology.Front Pharmacol. 2025 Jan 6;15:1499753. doi: 10.3389/fphar.2024.1499753. eCollection 2024. Front Pharmacol. 2025. PMID: 39834822 Free PMC article.
References
-
- Atsugi T., Yokouchi M., Hirano T., Hirabayashi A., Nagai T., Ohyama M., et al. . (2020). Holocrine secretion occurs outside the tight junction barrier in multicellular glands: lessons from Claudin-1-deficient mice. J. Invest. Dermatol. 140, 298–308.e5. doi: 10.1016/j.jid.2019.06.150, PMID: - DOI - PubMed
-
- Bai J. W., Deng W. W., Zhang J., Xu S. M., Zhang D. X. (2009). Protective effect of myosin light-chain kinase inhibitor on acute lung injury. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 21, 215–218, PMID: - PubMed
-
- Bücker R., Zakrzewski S. S., Wiegand S., Pieper R., Fromm A., Fromm M., et al. . (2020). Zinc prevents intestinal epithelial barrier dysfunction induced by alpha-hemolysin-producing Escherichia coli 536 infection in porcine colon. Vet. Microbiol. 243:108632. doi: 10.1016/j.vetmic.2020.108632, PMID: - DOI - PubMed
-
- Camara-Lemarroy C. R., Escobedo-Zúñiga N., Guzmán-de L. G. F., Castro-Garza J., Vargas-Villarreal J., Góngora-Rivera F. (2021). D-lactate and intestinal fatty acid-binding protein are elevated in serum in patients with acute ischemic stroke. Acta Neurol. Belg. 121, 87–93. doi: 10.1007/s13760-018-0940-x, PMID: - DOI - PubMed
LinkOut - more resources
Full Text Sources

