Repositioning of antiarrhythmics for prostate cancer treatment: a novel strategy to reprogram cancer-associated fibroblasts towards a tumor-suppressive phenotype
- PMID: 38858661
- PMCID: PMC11165820
- DOI: 10.1186/s13046-024-03081-0
Repositioning of antiarrhythmics for prostate cancer treatment: a novel strategy to reprogram cancer-associated fibroblasts towards a tumor-suppressive phenotype
Abstract
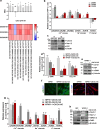
Background: Cancer-associated fibroblasts (CAFs) play a significant role in fueling prostate cancer (PCa) progression by interacting with tumor cells. A previous gene expression analysis revealed that CAFs up-regulate genes coding for voltage-gated cation channels, as compared to normal prostate fibroblasts (NPFs). In this study, we explored the impact of antiarrhythmic drugs, known cation channel inhibitors, on the activated state of CAFs and their interaction with PCa cells.
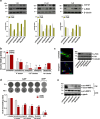
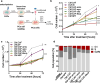
Methods: The effect of antiarrhythmic treatment on CAF activated phenotype was assessed in terms of cell morphology and fibroblast activation markers. CAF contractility and migration were evaluated by 3D gel collagen contraction and scratch assays, respectively. The ability of antiarrhythmics to impair CAF-PCa cell interplay was investigated in CAF-PCa cell co-cultures by assessing tumor cell growth and expression of epithelial-to-mesenchymal transition (EMT) markers. The effect on in vivo tumor growth was assessed by subcutaneously injecting PCa cells in SCID mice and intratumorally administering the medium of antiarrhythmic-treated CAFs or in co-injection experiments, where antiarrhythmic-treated CAFs were co-injected with PCa cells.
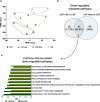
Results: Activated fibroblasts show increased membrane conductance for potassium, sodium and calcium, consistently with the mRNA and protein content analysis. Antiarrhythmics modulate the expression of fibroblast activation markers. Although to a variable extent, these drugs also reduce CAF motility and hinder their ability to remodel the extracellular matrix, for example by reducing MMP-2 release. Furthermore, conditioned medium and co-culture experiments showed that antiarrhythmics can, at least in part, reverse the protumor effects exerted by CAFs on PCa cell growth and plasticity, both in androgen-sensitive and castration-resistant cell lines. Consistently, the transcriptome of antiarrhythmic-treated CAFs resembles that of tumor-suppressive NPFs. In vivo experiments confirmed that the conditioned medium or the direct coinjection of antiarrhythmic-treated CAFs reduced the tumor growth rate of PCa xenografts.
Conclusions: Collectively, such data suggest a new therapeutic strategy for PCa based on the repositioning of antiarrhythmic drugs with the aim of normalizing CAF phenotype and creating a less permissive tumor microenvironment.
Keywords: Antiarrhythmics; Cancer-associated fibroblasts; Drug-repositioning; Prostate cancer.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Gandellini P, Andriani F, Merlino G, D’Aiuto F, Roz L, Callari M. Complexity in the tumour microenvironment: cancer associated fibroblast gene expression patterns identify both common and unique features of tumour-stroma crosstalk across cancer types. Semin Cancer Biol. 2015;35:96–106. doi: 10.1016/j.semcancer.2015.08.008. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

