Alterations in brain fluid physiology during the early stages of development of ischaemic oedema
- PMID: 38858667
- PMCID: PMC11163777
- DOI: 10.1186/s12987-024-00534-8
Alterations in brain fluid physiology during the early stages of development of ischaemic oedema
Abstract
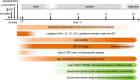
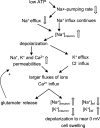
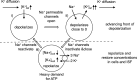
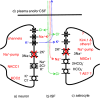
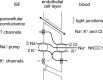
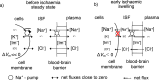
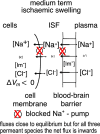
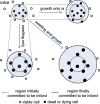
Oedema occurs when higher than normal amounts of solutes and water accumulate in tissues. In brain parenchymal tissue, vasogenic oedema arises from changes in blood-brain barrier permeability, e.g. in peritumoral oedema. Cytotoxic oedema arises from excess accumulation of solutes within cells, e.g. ischaemic oedema following stroke. This type of oedema is initiated when blood flow in the affected core region falls sufficiently to deprive brain cells of the ATP needed to maintain ion gradients. As a consequence, there is: depolarization of neurons; neural uptake of Na+ and Cl- and loss of K+; neuronal swelling; astrocytic uptake of Na+, K+ and anions; swelling of astrocytes; and reduction in ISF volume by fluid uptake into neurons and astrocytes. There is increased parenchymal solute content due to metabolic osmolyte production and solute influx from CSF and blood. The greatly increased [K+]isf triggers spreading depolarizations into the surrounding penumbra increasing metabolic load leading to increased size of the ischaemic core. Water enters the parenchyma primarily from blood, some passing into astrocyte endfeet via AQP4. In the medium term, e.g. after three hours, NaCl permeability and swelling rate increase with partial opening of tight junctions between blood-brain barrier endothelial cells and opening of SUR1-TPRM4 channels. Swelling is then driven by a Donnan-like effect. Longer term, there is gross failure of the blood-brain barrier. Oedema resolution is slower than its formation. Fluids without colloid, e.g. infused mock CSF, can be reabsorbed across the blood-brain barrier by a Starling-like mechanism whereas infused serum with its colloids must be removed by even slower extravascular means. Large scale oedema can increase intracranial pressure (ICP) sufficiently to cause fatal brain herniation. The potentially lethal increase in ICP can be avoided by craniectomy or by aspiration of the osmotically active infarcted region. However, the only satisfactory treatment resulting in retention of function is restoration of blood flow, providing this can be achieved relatively quickly. One important objective of current research is to find treatments that increase the time during which reperfusion is successful. Questions still to be resolved are discussed.
Keywords: ATP depletion; Aquaporin 4; Astrocyte swelling; Blood–brain barrier permeability; CSF influx; Donnan effect; Neuronal swelling; Perivascular spaces; Reperfusion; Spreading depolarization.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
The normal and pathological physiology of brain water.Adv Tech Stand Neurosurg. 1997;23:47-142. doi: 10.1007/978-3-7091-6549-2_2. Adv Tech Stand Neurosurg. 1997. PMID: 9075471 Review.
-
The study of the function of AQP4 in cerebral ischaemia-reperfusion injury using poroelastic theory.Int J Numer Method Biomed Eng. 2017 Jan;33(1). doi: 10.1002/cnm.2784. Epub 2016 Apr 22. Int J Numer Method Biomed Eng. 2017. PMID: 26991256
-
Pathophysiology of cerebral oedema in acute liver failure.World J Gastroenterol. 2013 Dec 28;19(48):9240-55. doi: 10.3748/wjg.v19.i48.9240. World J Gastroenterol. 2013. PMID: 24409052 Free PMC article. Review.
-
[Blood-brain barrier pathophysiology and ischaemic brain oedema].Ann Fr Anesth Reanim. 2003 Apr;22(4):312-9. doi: 10.1016/s0750-7658(03)00069-8. Ann Fr Anesth Reanim. 2003. PMID: 12818323 Review. French.
-
Fluid and ion transfer across the blood-brain and blood-cerebrospinal fluid barriers; a comparative account of mechanisms and roles.Fluids Barriers CNS. 2016 Oct 31;13(1):19. doi: 10.1186/s12987-016-0040-3. Fluids Barriers CNS. 2016. PMID: 27799072 Free PMC article. Review.
Cited by
-
Glymphatic dysfunction as a biomarker for post-stroke cognitive impairment.Sci Rep. 2025 Jun 3;15(1):19382. doi: 10.1038/s41598-025-04054-9. Sci Rep. 2025. PMID: 40461672 Free PMC article.
-
Regulation of brain fluid volumes and pressures: basic principles, intracranial hypertension, ventriculomegaly and hydrocephalus.Fluids Barriers CNS. 2024 Jul 17;21(1):57. doi: 10.1186/s12987-024-00532-w. Fluids Barriers CNS. 2024. PMID: 39020364 Free PMC article. Review.
-
Association between the postoperative glycemic variability and mortality after craniotomy: a retrospective cohort study and development of a mortality prediction model.Front Endocrinol (Lausanne). 2025 Jul 17;16:1613662. doi: 10.3389/fendo.2025.1613662. eCollection 2025. Front Endocrinol (Lausanne). 2025. PMID: 40747311 Free PMC article.
-
Electromechanical Regulation Underlying Protein Nanoparticle-Induced Osmotic Pressure in Neurotoxic Edema.Int J Nanomedicine. 2025 Apr 5;20:4145-4163. doi: 10.2147/IJN.S503181. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40207308 Free PMC article.
-
Brain fluid physiology in ischaemic stroke; more than just oedema.Fluids Barriers CNS. 2025 Jun 18;22(1):60. doi: 10.1186/s12987-025-00671-8. Fluids Barriers CNS. 2025. PMID: 40533800 Free PMC article. Review.
References
-
- Hladky SB, Barrand MA. Regulation of brain fluid volumes and pressures: basic principles, intracranial hypertension, ventriculomegaly and hydrocephalus Fluids Barriers CNS 2024. 10.1186/s12987-024-00532-w. (in press)
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

