Scoping review of clinical decision support systems for multiple sclerosis management: Leveraging information technology and massive health data
- PMID: 38860844
- PMCID: PMC11618115
- DOI: 10.1111/ene.16363
Scoping review of clinical decision support systems for multiple sclerosis management: Leveraging information technology and massive health data
Abstract
Background and purpose: Multiple sclerosis (MS) is a complex autoimmune disease of the central nervous system, with numerous therapeutic options, but a lack of biomarkers to support a mechanistic approach to precision medicine. A computational approach to precision medicine could proceed from clinical decision support systems (CDSSs). They are digital tools aiming to empower physicians through the clinical applications of information technology and massive data. However, the process of their clinical development is still maturing; we aimed to review it in the field of MS.
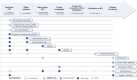
Methods: For this scoping review, we screened systematically the PubMed database. We identified 24 articles reporting 14 CDSS projects and compared their technical and software development aspects.
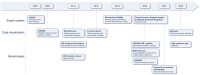
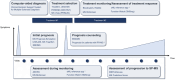
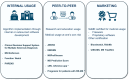
Results: The projects position themselves in various contexts of usage with various algorithmic approaches: expert systems, CDSSs based on similar patients' data visualization, and model-based CDSSs implementing mathematical predictive models. So far, no project has completed its clinical development up to certification for clinical use with global release. Some CDSSs have been replaced at subsequent project iterations. The most advanced projects did not necessarily report every step of clinical development in a dedicated article (proof of concept, offline validation, refined prototype, live clinical evaluation, comparative prospective evaluation). They seek different software distribution options to integrate into health care: internal usage, "peer-to-peer," and marketing distribution.
Conclusions: This review illustrates the potential of clinical applications of information technology and massive data to support MS management and helps clarify the roadmap for future projects as a multidisciplinary and multistep process.
Keywords: artificial intelligence; big data; clinical decision support system; multiple sclerosis; precision medicine.
© 2024 The Author(s). European Journal of Neurology published by John Wiley & Sons Ltd on behalf of European Academy of Neurology.
Conflict of interest statement
P.‐A.G. is the founder of Methodomics (2008) and the cofounder of Big Data Santé (2018). He consults for major pharmaceutical companies, all of which are handled through academic pipelines (AstraZeneca, Biogen, Boston Scientific, Cook, Edimark, Ellipses, Elsevier, Methodomics, Merck, Mérieux, Sanofi‐Genzyme, Octopize). P.‐A.G. is a volunteer board member at AXA not‐for‐profit mutual insurance company (2021). He has no prescription activity with either drugs or devices. None of the other authors has any conflict of interest to disclose.
Figures


Similar articles
-
Diagnosis of Rare Diseases: a scoping review of clinical decision support systems.Orphanet J Rare Dis. 2020 Sep 24;15(1):263. doi: 10.1186/s13023-020-01536-z. Orphanet J Rare Dis. 2020. PMID: 32972444 Free PMC article.
-
Applications of Clinical Decision Support Systems in Diabetes Care: Scoping Review.J Med Internet Res. 2023 Dec 8;25:e51024. doi: 10.2196/51024. J Med Internet Res. 2023. PMID: 38064249 Free PMC article.
-
Clinical decision support systems at the Vienna General Hospital using Arden Syntax: Design, implementation, and integration.Artif Intell Med. 2018 Nov;92:24-33. doi: 10.1016/j.artmed.2015.11.002. Epub 2015 Dec 1. Artif Intell Med. 2018. PMID: 26706047
-
What hinders the uptake of computerized decision support systems in hospitals? A qualitative study and framework for implementation.Implement Sci. 2017 Sep 15;12(1):113. doi: 10.1186/s13012-017-0644-2. Implement Sci. 2017. PMID: 28915822 Free PMC article.
-
Challenges and Facilitation Approaches for the Participatory Design of AI-Based Clinical Decision Support Systems: Protocol for a Scoping Review.JMIR Res Protoc. 2024 Sep 5;13:e58185. doi: 10.2196/58185. JMIR Res Protoc. 2024. PMID: 39235846 Free PMC article.
Cited by
-
Digital Representation of Patients as Medical Digital Twins: Data-Centric Viewpoint.JMIR Med Inform. 2025 Jan 28;13:e53542. doi: 10.2196/53542. JMIR Med Inform. 2025. PMID: 39881430 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

