Enzyme-independent role of EZH2 in regulating cell cycle progression via the SKP2-KIP/CIP pathway
- PMID: 38862595
- PMCID: PMC11166936
- DOI: 10.1038/s41598-024-64338-4
Enzyme-independent role of EZH2 in regulating cell cycle progression via the SKP2-KIP/CIP pathway
Erratum in
-
Author Correction: Enzyme-independent role of EZH2 in regulating cell cycle progression via the SKP2-KIP/CIP pathway.Sci Rep. 2024 Jul 27;14(1):17230. doi: 10.1038/s41598-024-68294-x. Sci Rep. 2024. PMID: 39060362 Free PMC article. No abstract available.
Abstract
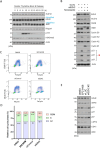
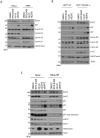
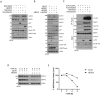
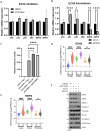
While EZH2 enzymatic activity is well-known, emerging evidence suggests that EZH2 can exert functions in a methyltransferase-independent manner. In this study, we have uncovered a novel mechanism by which EZH2 positively regulates the expression of SKP2, a critical protein involved in cell cycle progression. We demonstrate that depletion of EZH2 significantly reduces SKP2 protein levels in several cell types, while treatment with EPZ-6438, an EZH2 enzymatic inhibitor, has no effect on SKP2 protein levels. Consistently, EZH2 depletion leads to cell cycle arrest, accompanied by elevated expression of CIP/KIP family proteins, including p21, p27, and p57, whereas EPZ-6438 treatment does not modulate their levels. We also provide evidence that EZH2 knockdown, but not enzymatic inhibition, suppresses SKP2 mRNA expression, underscoring the transcriptional regulation of SKP2 by EZH2 in a methyltransferase-independent manner. Supporting this, analysis of the Cancer Genome Atlas database reveals a close association between EZH2 and SKP2 expression in human malignancies. Moreover, EZH2 depletion but not enzymatic inhibition positively regulates the expression of major epithelial-mesenchymal transition (EMT) regulators, such as ZEB1 and SNAIL1, in transformed cells. Our findings shed light on a novel mechanism by which EZH2 exerts regulatory effects on cell proliferation and differentiation through its methyltransferase-independent function, specifically by modulating SKP2 expression.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Cortactin modulates RhoA activation and expression of Cip/Kip cyclin-dependent kinase inhibitors to promote cell cycle progression in 11q13-amplified head and neck squamous cell carcinoma cells.Mol Cell Biol. 2010 Nov;30(21):5057-70. doi: 10.1128/MCB.00249-10. Epub 2010 Aug 30. Mol Cell Biol. 2010. PMID: 20805359 Free PMC article.
-
Focal adhesion kinase controls cellular levels of p27/Kip1 and p21/Cip1 through Skp2-dependent and -independent mechanisms.Mol Cell Biol. 2006 Jun;26(11):4201-13. doi: 10.1128/MCB.01612-05. Mol Cell Biol. 2006. PMID: 16705171 Free PMC article.
-
The NUCKS1-SKP2-p21/p27 axis controls S phase entry.Nat Commun. 2021 Nov 29;12(1):6959. doi: 10.1038/s41467-021-27124-8. Nat Commun. 2021. PMID: 34845229 Free PMC article.
-
Cellular Response upon Stress: p57 Contribution to the Final Outcome.Mediators Inflamm. 2015;2015:259325. doi: 10.1155/2015/259325. Epub 2015 Sep 27. Mediators Inflamm. 2015. PMID: 26491224 Free PMC article. Review.
-
The role of Skp2 and its substrate CDKN1B (p27) in colorectal cancer.J Gastrointestin Liver Dis. 2015 Jun;24(2):225-34. doi: 10.15403/jgld.2014.1121.242.skp2. J Gastrointestin Liver Dis. 2015. PMID: 26114183 Review.
Cited by
-
Topical MTH1 Inhibition Suppresses SKP2-WNT5a-Driven Psoriatic Hyperproliferation.Int J Mol Sci. 2025 Jul 25;26(15):7174. doi: 10.3390/ijms26157174. Int J Mol Sci. 2025. PMID: 40806307 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

