Tracking the evolution of anti-SARS-CoV-2 antibodies and long-term humoral immunity within 2 years after COVID-19 infection
- PMID: 38862731
- PMCID: PMC11167004
- DOI: 10.1038/s41598-024-64414-9
Tracking the evolution of anti-SARS-CoV-2 antibodies and long-term humoral immunity within 2 years after COVID-19 infection
Erratum in
-
Author Correction: Tracking the evolution of anti-SARS-CoV-2 antibodies and long-term humoral immunity within 2 years after COVID-19 infection.Sci Rep. 2024 Aug 22;14(1):19498. doi: 10.1038/s41598-024-70482-8. Sci Rep. 2024. PMID: 39174629 Free PMC article. No abstract available.
Abstract
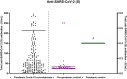
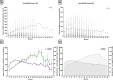
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that gave rise to COVID-19 infection produced a worldwide health crisis. The virus can cause a serious or even fatal disease. Comprehending the complex immunological responses triggered by SARS-CoV-2 infection is essential for identifying pivotal elements that shape the course of the disease and its enduring effects on immunity. The span and potency of antibody responses provide valuable perspicuity into the resilience of post-infection immunity. The analysis of existing literature reveals a diverse controversy, confining varying data about the persistence of particular antibodies as well as the multifaceted factors that impact their development and titer, Within this study we aimed to understand the dynamics of anti-SARS-CoV-2 antibodies against nucleocapsid (anti-SARS-CoV-2 (N)) and spike (anti-SARS-CoV-2 (N)) proteins in long-term immunity in convalescent patients, as well as the factors influencing the production and kinetics of those antibodies. We collected 6115 serum samples from 1611 convalescent patients at different post-infection intervals up to 21 months Study showed that in the fourth month, the anti-SARS-CoV-2 (N) exhibited their peak mean value, demonstrating a 79% increase compared to the initial month. Over the subsequent eight months, the peak value experienced a modest decline, maintaining a relatively elevated level by the end of study. Conversely, anti-SARS-CoV-2 (S) exhibited a consistent increase at each three-month interval over the 15-month period, culminating in a statistically significant peak mean value at the study's conclusion. Our findings demonstrate evidence of sustained seropositivity rates for both anti-SARS-CoV-2 (N) and (S), as well as distinct dynamics in the long-term antibody responses, with anti-SARS-CoV-2 (N) levels displaying remarkable persistence and anti-SARS-CoV-2 (S) antibodies exhibiting a progressive incline.
Keywords: Anti-SARS-CoV-2 (N); Anti-SARS-CoV-2 (S); Kinetics; Long-term humoral immunity.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests
Figures


Similar articles
-
SARS-CoV-2 Infection Severity Is Linked to Superior Humoral Immunity against the Spike.mBio. 2021 Jan 19;12(1):e02940-20. doi: 10.1128/mBio.02940-20. mBio. 2021. PMID: 33468695 Free PMC article.
-
Evolution of antibody responses up to 13 months after SARS-CoV-2 infection and risk of reinfection.EBioMedicine. 2021 Sep;71:103561. doi: 10.1016/j.ebiom.2021.103561. Epub 2021 Aug 27. EBioMedicine. 2021. PMID: 34455390 Free PMC article.
-
Longitudinal Profiling of Antibody Response in Patients With COVID-19 in a Tertiary Care Hospital in Beijing, China.Front Immunol. 2021 Mar 15;12:614436. doi: 10.3389/fimmu.2021.614436. eCollection 2021. Front Immunol. 2021. PMID: 33790892 Free PMC article.
-
Detrimental Effects of Anti-Nucleocapsid Antibodies in SARS-CoV-2 Infection, Reinfection, and the Post-Acute Sequelae of COVID-19.Pathogens. 2024 Dec 15;13(12):1109. doi: 10.3390/pathogens13121109. Pathogens. 2024. PMID: 39770368 Free PMC article. Review.
-
Obesity-compromised immunity in post-COVID-19 condition: a critical control point of chronicity.Front Immunol. 2024 Aug 12;15:1433531. doi: 10.3389/fimmu.2024.1433531. eCollection 2024. Front Immunol. 2024. PMID: 39188722 Free PMC article. Review.
Cited by
-
Magnetic bead-based electrochemical surrogate virus neutralization test for quantification of antibody neutralizing efficiency.Biosens Bioelectron. 2025 Nov 1;287:117640. doi: 10.1016/j.bios.2025.117640. Epub 2025 May 30. Biosens Bioelectron. 2025. PMID: 40554384 Free PMC article.
-
Exploring the Contrasts and Similarities of Dengue and SARS-CoV-2 Infections During the COVID-19 Era.Int J Mol Sci. 2024 Oct 29;25(21):11624. doi: 10.3390/ijms252111624. Int J Mol Sci. 2024. PMID: 39519178 Free PMC article. Review.
-
Kinetics of naturally induced binding and neutralising anti-SARS-CoV-2 antibody levels and potencies among SARS-CoV-2 infected Kenyans with diverse grades of COVID-19 severity: an observational study.Wellcome Open Res. 2024 Dec 2;8:350. doi: 10.12688/wellcomeopenres.19414.2. eCollection 2023. Wellcome Open Res. 2024. PMID: 39640868 Free PMC article.
-
Tracking the evolution of serum antibody levels and influencing factors post-SARS-CoV-2 infection among community residents in Fuzhou City.Front Immunol. 2025 Mar 31;16:1533102. doi: 10.3389/fimmu.2025.1533102. eCollection 2025. Front Immunol. 2025. PMID: 40230855 Free PMC article.
-
COVID-19 Seroprevalence in Romania: Insights from a Nationwide Antibody Study.Epidemiologia (Basel). 2025 Jun 4;6(2):26. doi: 10.3390/epidemiologia6020026. Epidemiologia (Basel). 2025. PMID: 40558600 Free PMC article.
References
-
- Cascella, M., Rajnik, M., Aleem, A., Dulebohn, S. C. & Di Napoli, R. Features, evaluation, and treatment of coronavirus (COVID-19). StatPearls (2024). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

