Gallnut tannic acid alleviates gut damage induced by Salmonella pullorum in broilers by enhancing barrier function and modulating microbiota
- PMID: 38863452
- PMCID: PMC11166010
- DOI: 10.3389/fvets.2024.1382288
Gallnut tannic acid alleviates gut damage induced by Salmonella pullorum in broilers by enhancing barrier function and modulating microbiota
Abstract
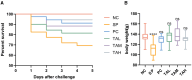
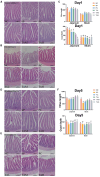
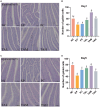
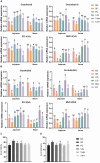
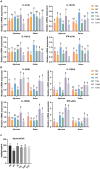
Pullorum disease (PD) is a bacterial infection caused by Salmonella pullorum (S. pullorum) that affects poultry. It is highly infectious and often fatal. Antibiotics are currently the mainstay of prophylactic and therapeutic treatments for PD, but their use can lead to the development of resistance in pathogenic bacteria and disruption of the host's intestinal flora. We added neomycin sulfate and different doses of tannic acid (TA) to the drinking water of chicks at 3 days of age and infected them with PD by intraperitoneal injection of S. pullorum at 9 days of age. We analyzed intestinal histopathological changes and the expression of immune-related genes and proteins by using the plate smear method, histological staining, real-time fluorescence quantitative PCR, ELISA kits, and 16S rRNA Analysis of intestinal flora. The results demonstrate that S. pullorum induces alterations in the immune status and impairs the functionality of the liver and intestinal barrier. We found that tannic acid significantly ameliorated S. pullorum-induced liver and intestinal damage, protected the intestinal physical and chemical barriers, restored the intestinal immune barrier function, and regulated the intestinal flora. Our results showed that TA has good anti-diarrhoeal, growth-promoting, immune-regulating, intestinal barrier-protecting and intestinal flora-balancing effects, and the best effect was achieved at an additive dose of 0.2%.
Keywords: Salmonella pullorum; gut barrier; intestinal flora; pullorum disease; tannic acid.
Copyright © 2024 Zou, Luan, Xi, Xue, Fan, Zhong, Zhou, Song, Zhao, Zou, Li, Jia, Fu, Liu and Yin.
Conflict of interest statement
HL was employed by Qilu Animal Health Products Co., Ltd. ZL was employed by Chengdu QianKun Veterinary Pharmaceutical Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

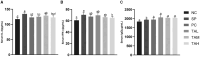

Similar articles
-
Bacillus coagulans MF-06 alleviates intestinal mucosal barrier from damage in chicks infected with Salmonella pullorum via activating the Wnt/β-catenin pathway.Front Microbiol. 2024 Nov 29;15:1492035. doi: 10.3389/fmicb.2024.1492035. eCollection 2024. Front Microbiol. 2024. PMID: 39678911 Free PMC article.
-
Lactobacillus casei protects intestinal mucosa from damage in chicks caused by Salmonella pullorum via regulating immunity and the Wnt signaling pathway and maintaining the abundance of gut microbiota.Poult Sci. 2021 Aug;100(8):101283. doi: 10.1016/j.psj.2021.101283. Epub 2021 May 28. Poult Sci. 2021. PMID: 34229217 Free PMC article.
-
Effects of a probiotic-fermented herbal blend on the growth performance, intestinal flora and immune function of chicks infected with Salmonella pullorum.Poult Sci. 2021 Jul;100(7):101196. doi: 10.1016/j.psj.2021.101196. Epub 2021 Apr 15. Poult Sci. 2021. PMID: 34111611 Free PMC article.
-
Plant-Derived Bioactive Compounds and Potential Health Benefits: Involvement of the Gut Microbiota and Its Metabolic Activity.Biomolecules. 2022 Dec 13;12(12):1871. doi: 10.3390/biom12121871. Biomolecules. 2022. PMID: 36551299 Free PMC article. Review.
-
Fowl typhoid and pullorum disease.Rev Sci Tech. 2000 Aug;19(2):405-24. doi: 10.20506/rst.19.2.1222. Rev Sci Tech. 2000. PMID: 10935271 Review.
Cited by
-
Bifidobacterium bifidum postbiotics prevent Salmonella Pullorum infection in chickens by modulating pyroptosis and enhancing gut health.Poult Sci. 2025 Apr;104(4):104968. doi: 10.1016/j.psj.2025.104968. Epub 2025 Mar 1. Poult Sci. 2025. PMID: 40043668 Free PMC article.
-
Effects of Chinese Gallotannins on Antioxidant Function, Intestinal Health, and Gut Flora in Broilers Challenged with Escherichia coli Lipopolysaccharide.Animals (Basel). 2024 Jun 28;14(13):1915. doi: 10.3390/ani14131915. Animals (Basel). 2024. PMID: 38998028 Free PMC article.
-
Bacillus coagulans MF-06 alleviates intestinal mucosal barrier from damage in chicks infected with Salmonella pullorum via activating the Wnt/β-catenin pathway.Front Microbiol. 2024 Nov 29;15:1492035. doi: 10.3389/fmicb.2024.1492035. eCollection 2024. Front Microbiol. 2024. PMID: 39678911 Free PMC article.
References
-
- Thøfner I, Christensen J. Bacterial Diseases in Poultry. Amsterdam: Elsevier; (2021), p. 199–227. 10.1016/B978-0-12-818030-3.00005-2 - DOI
-
- Wu QJ, Jiao C, Liu ZH, Li SW, Zhu DD, Ma WF, et al. . Effect of glutamine on the intestinal function and health of broilers challenged with Salmonella pullorum. Indian J Anim Res. (2019) 53:1210–6. 10.18805/ijar.B-1133 - DOI
LinkOut - more resources
Full Text Sources

