Non-invasive detection of orthotopic human lung tumors by microRNA expression profiling of mouse exhaled breath condensates and exhaled extracellular vesicles
- PMID: 38863869
- PMCID: PMC11165456
- DOI: 10.20517/evcna.2023.77
Non-invasive detection of orthotopic human lung tumors by microRNA expression profiling of mouse exhaled breath condensates and exhaled extracellular vesicles
Abstract
Aim: The lung is the second most frequent site of metastatic dissemination. Early detection is key to improving survival. Given that the lung interfaces with the external environment, the collection of exhaled breath condensate (EBC) provides the opportunity to obtain biological material including exhaled miRNAs that originate from the lung.
Methods: In this proof-of-principal study, we used the highly metastatic MDA-MB-231 subline 3475 breast cancer cell line (LM-3475) to establish an orthotopic lung tumor-bearing mouse model and investigate non-invasive detection of lung tumors by analysis of exhaled miRNAs. We initially conducted miRNA NGS and qPCR validation analyses on condensates collected from unrestrained animals and identified significant miRNA expression differences between the condensates of lung tumor-bearing and control mice. To focus our purification of EBC and evaluate the origin of these differentially expressed miRNAs, we developed a system to collect EBC directly from the nose and mouth of our mice.
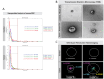
Results: Using nanoparticle distribution analyses, TEM, and ONi super-resolution nanoimaging, we determined that human tumor EVs could be increasingly detected in mouse EBC during the progression of secondary lung tumors. Using our customizable EV-CATCHER assay, we purified human tumor EVs from mouse EBC and demonstrated that the bulk of differentially expressed exhaled miRNAs originate from lung tumors, which could be detected by qPCR within 1 to 2 weeks after tail vein injection of the metastatic cells.
Conclusion: This study is the first of its kind and demonstrates that lung tumor EVs are exhaled in mice and provide non-invasive biomarkers for detection of lung tumors.
Keywords: Extracellular vesicles; exhaled breath condensate; miRNAs; orthotopic lung tumor-bearing animal model.
Conflict of interest statement
Conflicts of Interest Olivier Loudig is a Junior Editorial Board member of the journal Extracellular Vesicles and Circulating Nucleic Acids. The other authors declared that there are no conflicts of interest.
Figures






Similar articles
-
Current Evidence Supporting the Role of miRNA as a Biomarker for Lung Cancer Diagnosis Through Exhaled Breath Condensate Collection: A Narrative Review.Life (Basel). 2025 Apr 22;15(5):683. doi: 10.3390/life15050683. Life (Basel). 2025. PMID: 40430112 Free PMC article. Review.
-
Alterations in Abundance and Compartmentalization of miRNAs in Blood Plasma Extracellular Vesicles and Extracellular Condensates during HIV/SIV Infection and Its Modulation by Antiretroviral Therapy (ART) and Delta-9-Tetrahydrocannabinol (Δ9-THC).Viruses. 2023 Feb 24;15(3):623. doi: 10.3390/v15030623. Viruses. 2023. PMID: 36992332 Free PMC article.
-
MicroRNAs in exhaled breath condensate: A pilot study of biomarker detection for lung cancer.Cancer Treat Res Commun. 2023;35:100689. doi: 10.1016/j.ctarc.2023.100689. Epub 2023 Feb 10. Cancer Treat Res Commun. 2023. PMID: 36773435
-
Clinical Utility of microRNAs in Exhaled Breath Condensate as Biomarkers for Lung Cancer.J Pers Med. 2021 Feb 9;11(2):111. doi: 10.3390/jpm11020111. J Pers Med. 2021. PMID: 33572343 Free PMC article.
-
Clinical Application of Exhaled Breath Condensate in Diagnosis of Diseases.Discov Med. 2024 Jun;36(185):1154-1161. doi: 10.24976/Discov.Med.202436185.105. Discov Med. 2024. PMID: 38926101 Review.
Cited by
-
Customizing EV-CATCHER to Purify Placental Extracellular Vesicles from Maternal Plasma to Detect Placental Pathologies.Int J Mol Sci. 2024 May 7;25(10):5102. doi: 10.3390/ijms25105102. Int J Mol Sci. 2024. PMID: 38791142 Free PMC article.
-
Ultrasensitive cDNA Library Preparation for Next-generation Sequencing of MicroRNAs from Small Extracellular Vesicles.J Vis Exp. 2025 Jun 13;(220):10.3791/67154. doi: 10.3791/67154. J Vis Exp. 2025. PMID: 40587398
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
