Modeling US blood donor deferrals under a policy of individual risk assessment for HIV risk sexual behavior
- PMID: 38864291
- PMCID: PMC11328673
- DOI: 10.1111/trf.17916
Modeling US blood donor deferrals under a policy of individual risk assessment for HIV risk sexual behavior
Abstract
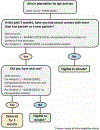
Background: In May 2023, the Food and Drug Administration (FDA) released final guidance for blood donor eligibility that recommended the elimination of 3-month deferral for men who have sex with men (MSM) and the related deferral for women who have sex with MSM. In its place, FDA introduced an individual risk assessment policy of asking all presenting blood donors, regardless of sex or gender, if they have had a new partner or more than one sexual partner in the last 3 months and deferring those who also report anal sex (penile-anal intercourse) during this period. We modeled the possible impact of this policy on the US blood donor base.
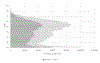
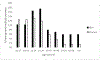
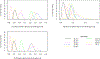
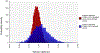
Study design and methods: We developed a computational model to estimate the percentage of blood donors who would be deferred under a policy of individual HIV risk assessment. The model incorporated demographic information about donors and national survey data on HIV risk behaviors and included age and sex distributions and dependencies.
Results: Our model estimates that approximately 1.2% of US blood donors would be deferred under the individual HIV risk assessment paradigm.
Discussion: The model predicts a relatively minor effect of replacing the time-based deferral for MSM with individual risk-based deferral for sexual behavior. As US blood centers implement this new policy, the effect may be mitigated by donor gains, which warrant further study. The new policy is unlikely to adversely affect the availability of blood and blood components.
Keywords: HIV risk assessment; MSM deferral; anal sex; blood donor deferral; blood donor loss; modeling; sexual partner; transfusion.
© 2024 AABB. This article has been contributed to by U.S. Government employees and their work is in the public domain in the USA.
Conflict of interest statement
Conflict of interest:
No conflict of interest declared.
Figures
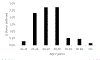
Similar articles
-
Behavioral interventions to reduce risk for sexual transmission of HIV among men who have sex with men.Cochrane Database Syst Rev. 2008 Jul 16;(3):CD001230. doi: 10.1002/14651858.CD001230.pub2. Cochrane Database Syst Rev. 2008. PMID: 18646068
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
-
Reinstatement and infectious disease rates among blood donors previously deferred under men who have sex with men time-based deferral.Transfusion. 2025 Aug;65(8):1472-1483. doi: 10.1111/trf.18299. Epub 2025 Jun 11. Transfusion. 2025. PMID: 40497820
-
Men who have sex with men and risk for transfusion-transmissible infections in blood donors in Western countries: A systematic review update.Vox Sang. 2023 Sep;118(9):709-720. doi: 10.1111/vox.13482. Epub 2023 Jun 15. Vox Sang. 2023. PMID: 37322809
-
Analysis of the most common causes of blood donor deferral in Southern Mozambique.BMC Infect Dis. 2025 Aug 7;25(1):994. doi: 10.1186/s12879-025-11393-5. BMC Infect Dis. 2025. PMID: 40775295 Free PMC article.
References
-
- Institute of Medicine Committee to Study HIV Transmission Through Blood. HIV and the Blood Supply: An Analysis of Crisis Decisionmaking. In: Leveton LB, Sox HC Jr., Stoto MA, eds. HIV and the Blood Supply: An Analysis of Crisis Decisionmaking. Washington (DC): National Academies Press (US), 1995. - PubMed
-
- CDC. Human immunodeficiency virus infection in transfusion recipients and their family members. MMWR Morb Mortal Wkly Rep 1987;36: 137–40. - PubMed
-
- CDC. Possible transfusion-associated acquired immune deficiency syndrome (AIDS) - California . MMWR Morb Mortal Wkly Rep 1982;31: 652–4. - PubMed
-
- CDC. Update: revised Public Health Service definition of persons who should refrain from donating blood and plasma--United States. MMWR Morb Mortal Wkly Rep 1985;34: 547–8. - PubMed
-
- FDA. Recommendations to Decrease the Risk of Transmitting Acquired Immune Deficiency Syndrome (AIDS) from Blood Donors, March 24, 1983.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

