Electroanatomical adaptations in the guinea pig heart from neonatal to adulthood
- PMID: 38864516
- PMCID: PMC11218563
- DOI: 10.1093/europace/euae158
Electroanatomical adaptations in the guinea pig heart from neonatal to adulthood
Abstract
Aims: Electroanatomical adaptations during the neonatal to adult phase have not been comprehensively studied in preclinical animal models. To explore the impact of age as a biological variable on cardiac electrophysiology, we employed neonatal and adult guinea pigs, which are a recognized animal model for developmental research.
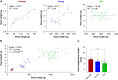
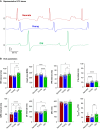
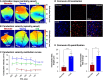
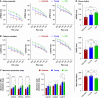
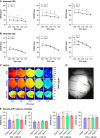
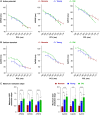
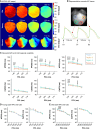
Methods and results: Electrocardiogram recordings were collected in vivo from anaesthetized animals. A Langendorff-perfusion system was employed for the optical assessment of action potentials and calcium transients. Optical data sets were analysed using Kairosight 3.0 software. The allometric relationship between heart weight and body weight diminishes with age, it is strongest at the neonatal stage (R2 = 0.84) and abolished in older adults (R2 = 1E-06). Neonatal hearts exhibit circular activation, while adults show prototypical elliptical shapes. Neonatal conduction velocity (40.6 ± 4.0 cm/s) is slower than adults (younger: 61.6 ± 9.3 cm/s; older: 53.6 ± 9.2 cm/s). Neonatal hearts have a longer action potential duration (APD) and exhibit regional heterogeneity (left apex; APD30: 68.6 ± 5.6 ms, left basal; APD30: 62.8 ± 3.6), which was absent in adults. With dynamic pacing, neonatal hearts exhibit a flatter APD restitution slope (APD70: 0.29 ± 0.04) compared with older adults (0.49 ± 0.04). Similar restitution characteristics are observed with extrasystolic pacing, with a flatter slope in neonates (APD70: 0.54 ± 0.1) compared with adults (younger: 0.85 ± 0.4; older: 0.95 ± 0.7). Neonatal hearts display unidirectional excitation-contraction coupling, while adults exhibit bidirectionality.
Conclusion: Postnatal development is characterized by transient changes in electroanatomical properties. Age-specific patterns can influence cardiac physiology, pathology, and therapies for cardiovascular diseases. Understanding heart development is crucial to evaluating therapeutic eligibility, safety, and efficacy.
Keywords: Cardiac electrophysiology; Developmental biology; Optical mapping; Pediatric models.
© The Author(s) 2024. Published by Oxford University Press on behalf of the European Society of Cardiology.
Conflict of interest statement
Conflict of interest: None declared.
Figures
Update of
-
Electroanatomical Adaptations in the Guinea Pig Heart from Neonatal to Adulthood.bioRxiv [Preprint]. 2024 Jan 30:2024.01.26.577234. doi: 10.1101/2024.01.26.577234. bioRxiv. 2024. Update in: Europace. 2024 Jul 2;26(7):euae158. doi: 10.1093/europace/euae158. PMID: 38352347 Free PMC article. Updated. Preprint.
Similar articles
-
Electroanatomical Adaptations in the Guinea Pig Heart from Neonatal to Adulthood.bioRxiv [Preprint]. 2024 Jan 30:2024.01.26.577234. doi: 10.1101/2024.01.26.577234. bioRxiv. 2024. Update in: Europace. 2024 Jul 2;26(7):euae158. doi: 10.1093/europace/euae158. PMID: 38352347 Free PMC article. Updated. Preprint.
-
Age-dependent changes in electrophysiology and calcium handling: implications for pediatric cardiac research.Am J Physiol Heart Circ Physiol. 2020 Feb 1;318(2):H354-H365. doi: 10.1152/ajpheart.00521.2019. Epub 2019 Dec 30. Am J Physiol Heart Circ Physiol. 2020. PMID: 31886723 Free PMC article.
-
The effect of sex and age on ex vivo cardiac electrophysiology: insight from a guinea pig model.Am J Physiol Heart Circ Physiol. 2023 Jan 1;324(1):H141-H154. doi: 10.1152/ajpheart.00497.2022. Epub 2022 Dec 9. Am J Physiol Heart Circ Physiol. 2023. PMID: 36487188 Free PMC article.
-
In vivo ratiometric optical mapping enables high-resolution cardiac electrophysiology in pig models.Cardiovasc Res. 2019 Sep 1;115(11):1659-1671. doi: 10.1093/cvr/cvz039. Cardiovasc Res. 2019. PMID: 30753358 Free PMC article.
-
The Zebrafish Heart as a Model of Mammalian Cardiac Function.Rev Physiol Biochem Pharmacol. 2016;171:99-136. doi: 10.1007/112_2016_5. Rev Physiol Biochem Pharmacol. 2016. PMID: 27538987 Review.
Cited by
-
Small Extracellular Vesicles from Breast Cancer Cells Induce Cardiotoxicity.Int J Mol Sci. 2025 Jan 23;26(3):945. doi: 10.3390/ijms26030945. Int J Mol Sci. 2025. PMID: 39940718 Free PMC article.
References
-
- Packer M, Carver JR, Rodeheffer RJ, Ivanhoe RJ, DiBianco R, Zeldis SM et al. Effect of oral milrinone on mortality in severe chronic heart failure. The PROMISE study research group. N Engl J Med 1991;325:1468–75. - PubMed
-
- Shipley JB, Tolman D, Hastillo A, Hess ML. Milrinone: basic and clinical pharmacology and acute and chronic management. Am J Med Sci Elsevier 1996;311:286–91. - PubMed
-
- Lue HC, Wu MH, Wang JK, Lin MT, Lu CW, Chiu SN et al. Normal ECG standards and percentile charts for infants, children and adolescents. Pediatr Neonatol 2023;64:256–73. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

