Glycodeoxycholic Acid Inhibits Primary Bile Acid Synthesis With Minor Effects on Glucose and Lipid Homeostasis in Humans
- PMID: 38864544
- PMCID: PMC12012696
- DOI: 10.1210/clinem/dgae399
Glycodeoxycholic Acid Inhibits Primary Bile Acid Synthesis With Minor Effects on Glucose and Lipid Homeostasis in Humans
Abstract
Background: Bile acids play vital roles in control of lipid, glucose, and energy metabolism by activating Takeda G protein-coupled receptor 5 and Farnesoid X receptor, the latter promoting production of the endocrine-acting fibroblast growth factor 19 (FGF19). Short-term administration of single bile acids has been reported to enhance plasma levels of GLP-1 and to enhance energy expenditure. However, prolonged bile acid supplementation (eg, of chenodeoxycholic acid for gallstone dissolution) has been reported to have adverse effects.
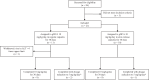
Study design: In this proof-of-concept study, we assessed the safety and metabolic effects of oral glycine-conjugated deoxycholic acid (GDCA) administration at 10 mg/kg/day using regular and slow-release capsules (mimicking physiological bile acid release) over 30 days in 2 groups of each 10 healthy lean men, respectively.
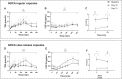
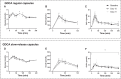
Main findings: GDCA increased postprandial total bile acid and FGF19 concentrations while suppressing those of the primary bile acids chenodeoxycholic acid and cholic acid. Plasma levels of 7α-hydroxy-4-cholesten-3-one were reduced, indicating repressed hepatic bile acid synthesis. There were minimal effects on indices of lipid, glucose, and energy metabolism. No serious adverse events were reported during GDCA administration in either capsule types, although 50% of participants showed mild increases in plasma levels of liver transaminases and 80% (regular capsules) and 50% (slow-release capsules) of participants experienced gastrointestinal adverse events.
Conclusion: GDCA administration leads to elevated FGF19 levels and effectively inhibits primary bile acid synthesis, supporting therapy compliance and its effectiveness. However, effects on lipid, glucose, and energy metabolism were minimal, indicating that expanding the pool of this relatively hydrophobic bile acid does not impact energy metabolism in healthy subjects.
Keywords: Farnesoid X receptor; Takeda G protein-coupled receptor 5; bile acids; glucagon-like peptide 1; glycodeoxycholic acid; humans; metabolic diseases.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Endocrine Society.
Figures
Similar articles
-
Differential effects of a 40-hour fast and bile acid supplementation on human GLP-1 and FGF19 responses.Am J Physiol Endocrinol Metab. 2019 Sep 1;317(3):E494-E502. doi: 10.1152/ajpendo.00534.2018. Epub 2019 Jun 25. Am J Physiol Endocrinol Metab. 2019. PMID: 31237451 Clinical Trial.
-
Obeticholic acid may increase the risk of gallstone formation in susceptible patients.J Hepatol. 2019 Nov;71(5):986-991. doi: 10.1016/j.jhep.2019.06.011. Epub 2019 Jun 27. J Hepatol. 2019. PMID: 31254596 Clinical Trial.
-
Prolonged fibroblast growth factor 19 response in patients with primary sclerosing cholangitis after an oral chenodeoxycholic acid challenge.Hepatol Int. 2017 Jan;11(1):132-140. doi: 10.1007/s12072-016-9769-7. Epub 2016 Sep 30. Hepatol Int. 2017. PMID: 27696157 Free PMC article.
-
Bile acids are nutrient signaling hormones.Steroids. 2014 Aug;86:62-8. doi: 10.1016/j.steroids.2014.04.016. Epub 2014 May 10. Steroids. 2014. PMID: 24819989 Free PMC article. Review.
-
Beyond intestinal soap--bile acids in metabolic control.Nat Rev Endocrinol. 2014 Aug;10(8):488-98. doi: 10.1038/nrendo.2014.60. Epub 2014 May 13. Nat Rev Endocrinol. 2014. PMID: 24821328 Review.
References
-
- Kuipers F, Bloks VW, Groen AK. Beyond intestinal soap–bile acids in metabolic control. Nat Rev Endocrinol. 2014;10(8):488‐498. - PubMed
-
- Schaap FG, Trauner M, Jansen PLM. Bile acid receptors as targets for drug development. Nat Rev Gastroenterol Hepatol. 2014;11(1):55‐67. - PubMed
-
- Sato H, Macchiarulo A, Thomas C, et al. . Novel potent and selective bile acid derivatives as TGR5 agonists: biological screening, structure-activity relationships, and molecular modeling studies. J Med Chem. 2008;51(6):1831‐1841. - PubMed
-
- Ridlon JM, Kang DJ, Hylemon PB. Bile salt biotransformations by human intestinal bacteria. J Lipid Res. 2006;47(2):241‐259. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

