Diversity of major histocompatibility complex of II B gene and mate choice in a monogamous and long-lived seabird, the Little Auk (Alle alle)
- PMID: 38865310
- PMCID: PMC11168636
- DOI: 10.1371/journal.pone.0304275
Diversity of major histocompatibility complex of II B gene and mate choice in a monogamous and long-lived seabird, the Little Auk (Alle alle)
Abstract
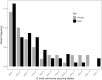
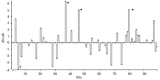
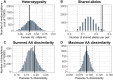
The major histocompatibility complex (MHC) plays a key role in the adaptive immune system of vertebrates, and is known to influence mate choice in many species. In birds, the MHC has been extensively examined but mainly in galliforms and passerines while other taxa that represent specific ecological and evolutionary life-histories, like seabirds, are underexamined. Here, we characterized diversity of MHC Class II B exon 2 in a colonial pelagic seabird, the Little Auk (or Dovekie Alle alle). We further examined whether MHC variation could be maintained through balancing selection and disassortative mating. We found high polymorphism at the genotyped MHC fragment, characterizing 99 distinct alleles across 140 individuals from three populations. The alleles frequencies exhibited a similar skewed distribution in both sexes, with the four most commonly occurring alleles representing approximately 35% of allelic variation. The results of a Bayesian site-by-site selection analysis suggest evidence of balancing selection and no direct evidence for MHC-dependent disassortative mating preferences in the Little Auk. The latter result might be attributed to the high overall polymorphism of the examined fragment, which itself may be maintained by the large population size of the species.
Copyright: © 2024 Wojczulanis-Jakubas et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Allelic diversity and patterns of selection at the major histocompatibility complex class I and II loci in a threatened shorebird, the Snowy Plover (Charadrius nivosus).BMC Evol Biol. 2020 Sep 10;20(1):114. doi: 10.1186/s12862-020-01676-7. BMC Evol Biol. 2020. PMID: 32912143 Free PMC article.
-
Gene duplication and divergence produce divergent MHC genotypes without disassortative mating.Mol Ecol. 2016 Sep;25(17):4355-67. doi: 10.1111/mec.13747. Epub 2016 Jul 29. Mol Ecol. 2016. PMID: 27376487 Free PMC article.
-
Characterization of MHC class II B polymorphism in bottlenecked New Zealand saddlebacks reveals low levels of genetic diversity.Immunogenetics. 2013 Aug;65(8):619-33. doi: 10.1007/s00251-013-0708-7. Epub 2013 May 18. Immunogenetics. 2013. PMID: 23686447
-
Meta-analysis of major histocompatibility complex (MHC) class IIA reveals polymorphism and positive selection in many vertebrate species.Mol Ecol. 2022 Dec;31(24):6390-6406. doi: 10.1111/mec.16726. Epub 2022 Oct 19. Mol Ecol. 2022. PMID: 36208104 Free PMC article. Review.
-
A quantitative review of MHC-based mating preference: the role of diversity and dissimilarity.Mol Ecol. 2014 Nov;23(21):5151-63. doi: 10.1111/mec.12934. Epub 2014 Oct 8. Mol Ecol. 2014. PMID: 25251264 Review.
References
-
- Murphy K., Travers P, Walport M. Janeway’s immunobiology. 7th ed. London: Tylor & Francis; 2008.
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

