Iron metabolism and arthritis: Exploring connections and therapeutic avenues
- PMID: 38867424
- PMCID: PMC11268821
- DOI: 10.1097/CM9.0000000000003169
Iron metabolism and arthritis: Exploring connections and therapeutic avenues
Abstract
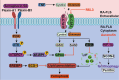
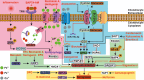
Iron is indispensable for the viablility of nearly all living organisms, and it is imperative for cells, tissues, and organisms to acquire this essential metal sufficiently and maintain its metabolic stability for survival. Disruption of iron homeostasis can lead to the development of various diseases. There is a robust connection between iron metabolism and infection, immunity, inflammation, and aging, suggesting that disorders in iron metabolism may contribute to the pathogenesis of arthritis. Numerous studies have focused on the significant role of iron metabolism in the development of arthritis and its potential for targeted drug therapy. Targeting iron metabolism offers a promising approach for individualized treatment of arthritis. Therefore, this review aimed to investigate the mechanisms by which the body maintains iron metabolism and the impacts of iron and iron metabolism disorders on arthritis. Furthermore, this review aimed to identify potential therapeutic targets and active substances related to iron metabolism, which could provide promising research directions in this field.
Copyright © 2024 The Chinese Medical Association, produced by Wolters Kluwer, Inc. under the CC-BY-NC-ND license.
Conflict of interest statement
None.
Figures

Similar articles
-
Iron Metabolism in Aging and Age-Related Diseases.Int J Mol Sci. 2022 Mar 25;23(7):3612. doi: 10.3390/ijms23073612. Int J Mol Sci. 2022. PMID: 35408967 Free PMC article. Review.
-
Alterations in Cellular Iron Metabolism Provide More Therapeutic Opportunities for Cancer.Int J Mol Sci. 2018 May 22;19(5):1545. doi: 10.3390/ijms19051545. Int J Mol Sci. 2018. PMID: 29789480 Free PMC article. Review.
-
Disruption of Iron Homeostasis and Mitochondrial Metabolism Are Promising Targets to Inhibit Candida auris.Microbiol Spectr. 2022 Apr 27;10(2):e0010022. doi: 10.1128/spectrum.00100-22. Epub 2022 Apr 12. Microbiol Spectr. 2022. PMID: 35412372 Free PMC article.
-
The Impacts of Iron Overload and Ferroptosis on Intestinal Mucosal Homeostasis and Inflammation.Int J Mol Sci. 2022 Nov 17;23(22):14195. doi: 10.3390/ijms232214195. Int J Mol Sci. 2022. PMID: 36430673 Free PMC article. Review.
-
Effects of adjuvant arthritis on copper, zinc, and iron metabolism in the rat.Res Commun Chem Pathol Pharmacol. 1989 Jan;63(1):153-6. Res Commun Chem Pathol Pharmacol. 1989. PMID: 2916078
Cited by
-
Enhanced cytotoxicity of aging associated NKG2C + CD8 + T cells in ankylosing spondylitis via HLA-B27.Arthritis Res Ther. 2025 Jun 14;27(1):125. doi: 10.1186/s13075-025-03585-w. Arthritis Res Ther. 2025. PMID: 40517268 Free PMC article.
-
Single-cell transcriptome and multi-omics integration reveal ferroptosis-driven immune microenvironment remodeling in knee osteoarthritis.Front Immunol. 2025 Jun 25;16:1608378. doi: 10.3389/fimmu.2025.1608378. eCollection 2025. Front Immunol. 2025. PMID: 40636124 Free PMC article.
-
Association of physical activity, cardiorespiratory fitness, grip strength, and grip strength asymmetry with incident musculoskeletal disorders in 406,080 White adults.J Sport Health Sci. 2025 Apr 8;14:101040. doi: 10.1016/j.jshs.2025.101040. Online ahead of print. J Sport Health Sci. 2025. PMID: 40210151 Free PMC article.
References
-
- Dougados M, Baeten D. Spondyloarthritis. Lancet 2011;377:2127–2137. doi: 10.1016/s0140-6736(11)60071-8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

