Invasive Pneumococcal Disease Epidemiology and Serotype Replacement After the Introduction of the 13-Valent Pneumococcal Conjugate Vaccine in Ontario, Canada, 2007-2022
- PMID: 38868312
- PMCID: PMC11167672
- DOI: 10.1093/ofid/ofae275
Invasive Pneumococcal Disease Epidemiology and Serotype Replacement After the Introduction of the 13-Valent Pneumococcal Conjugate Vaccine in Ontario, Canada, 2007-2022
Abstract
Background: New vaccine products were recently authorized for protection against invasive pneumococcal disease (IPD) in Canada. Our aim was to determine age- and serotype-specific trends in IPD incidence and severity in Canada's largest province, Ontario.
Methods: We included all confirmed IPD cases reported in Ontario and defined the pre-pneumococcal 13-valent conjugate vaccine (PCV13) era (01/2007 to 12/2010), post-PCV13 era (01/2011 to 12/2019), and coronavirus disease 2019 (COVID-19) pandemic era (01/2020 to 12/2022). We estimated incidence, hospitalization, and case fatality rate (CFR) by age. We grouped IPD cases by vaccine-specific serotypes (PCV13; PCV15-non-PCV13; PCV20-non-PCV13; PCV20-non-PCV15; polysaccharide 23-valent vaccine-non-PCV20; and non-vaccine-preventable [NVP]). We then compared incidence rates by age and serotype group in the pre- and post-PCV13 eras by calculating rate ratios (RRs) and their 95% CIs.
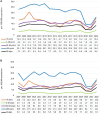
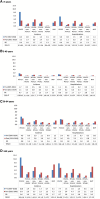
Results: Incidence and hospitalizations declined from the pre- to post-PCV13 era in children aged <5 years (RR, 0.7; 95% CI, 0.6-0.8; and RR, 0.8; 95% CI, 0.7-0.9, respectively), but the CFR increased (1.4% to 2.3%). Other age groups saw smaller declines or more stable incidence rates across the years; hospitalizations increased in adults aged 50-64 years (RR, 1.2; 95% CI, 1.1-1.4) and ≥65 years (RR, 1.1; 95% CI, 1.0-1.1). For all ages, IPD cases and hospitalizations attributable to PCV13 serotypes declined, and those attributable to PCV15-non-PCV13, PCV20-non-PCV13, and NVP serotypes increased. IPD incidence declined during the COVID-19 era.
Conclusions: IPD incidence and hospitalizations due to PCV13 serotypes decreased after PCV13 introduction but increased for other serotypes. Continued surveillance is required to evaluate changes to pneumococcal vaccination programs and ongoing changes to the distribution of IPD-causing serotypes.
Keywords: Invasive pneumococcal disease; epidemiology; serotype replacement; vaccination program.
© The Author(s) 2024. Published by Oxford University Press on behalf of Infectious Diseases Society of America.
Conflict of interest statement
Potential conflicts of interest. All authors: no reported conflicts.
Figures
Similar articles
-
Invasive pneumococcal disease in Canada 2010-2017: The role of current and next-generation higher-valent pneumococcal conjugate vaccines.Vaccine. 2021 May 21;39(22):3007-3017. doi: 10.1016/j.vaccine.2021.02.069. Epub 2021 Apr 3. Vaccine. 2021. PMID: 33824041
-
Historical Population-Level Impact of Infant 13-Valent Pneumococcal Conjugate Vaccine (PCV13) National Immunization Programs on Invasive Pneumococcal Disease in Australia, Canada, England and Wales, Israel, and the United States.Infect Dis Ther. 2023 May;12(5):1351-1364. doi: 10.1007/s40121-023-00798-x. Epub 2023 Apr 20. Infect Dis Ther. 2023. PMID: 37079175 Free PMC article.
-
PCV13-Serotype Breakthrough Pneumococcal Disease in Infants Receiving High-Valency Conjugate Vaccines: Population-Level Modeling in France.Infect Dis Ther. 2025 Apr;14(4):753-764. doi: 10.1007/s40121-025-01123-4. Epub 2025 Mar 19. Infect Dis Ther. 2025. PMID: 40106179 Free PMC article.
-
The 20-valent pneumococcal conjugate vaccine (PCV20): expected added value.Acta Clin Belg. 2023 Feb;78(1):78-86. doi: 10.1080/17843286.2022.2039865. Epub 2022 Feb 16. Acta Clin Belg. 2023. PMID: 35171752 Review.
-
20-Valent Pneumococcal Conjugate Vaccine: A Review of Its Use in Adults.Drugs. 2022 Jun;82(9):989-999. doi: 10.1007/s40265-022-01733-z. Epub 2022 Jul 6. Drugs. 2022. PMID: 35793027 Review.
Cited by
-
Burden of invasive pneumococcal disease, non-invasive all-cause pneumonia, and acute otitis media in hospitalized US children: a retrospective multi-center study from 2015 to 2020.BMC Health Serv Res. 2024 Dec 18;24(1):1574. doi: 10.1186/s12913-024-11898-w. BMC Health Serv Res. 2024. PMID: 39696329 Free PMC article.
-
Cost effectiveness of a 21-valent pneumococcal conjugate vaccine in adults: A systematic review of economic evaluations.Can Commun Dis Rep. 2025 Feb 12;51(2-3):84-91. doi: 10.14745/ccdr.v51i23a03. eCollection 2025 Feb. Can Commun Dis Rep. 2025. PMID: 39980572 Free PMC article.
-
The microbiological characteristics and diagnosis of Streptococcus pneumoniae infection in the conjugate vaccine era.Hum Vaccin Immunother. 2025 Dec;21(1):2497611. doi: 10.1080/21645515.2025.2497611. Epub 2025 Apr 27. Hum Vaccin Immunother. 2025. PMID: 40289536 Free PMC article. Review.
References
-
- O’Brien KL, Wolfson LJ, Watt JP, et al. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates. Lancet 2009; 374:893–902. - PubMed
-
- Drijkoningen JJC, Rohde GGU. Pneumococcal infection in adults: burden of disease. Clin Microbiol Infect 2014; 20(S5):45–51. - PubMed
-
- Troeger C, Blacker B, Khalil IA, et al. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory infections in 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect Dis 2018; 18:1191–210. - PMC - PubMed
-
- Government of Ontario . Ontario demographic quarterly: highlights of third quarter. 2024. Available at: http://www.ontario.ca/page/ontario-demographic-quarterly-highlights-thir.... Accessed March 27, 2024.
-
- Ontario Ministry of Health and Long-Term Care . Publicly Funded Immunization Schedules for Ontario June 2022. Ontario Ministry of Health; 2022.

