Human V 7.2-J 33 mucosal-associated invariant T cells in endometrial ectopic tissues tend to produce interferon-gamma: A new player in endometriosis etiology: A case-control study
- PMID: 38868448
- PMCID: PMC11165224
- DOI: 10.18502/ijrm.v22i3.16168
Human V 7.2-J 33 mucosal-associated invariant T cells in endometrial ectopic tissues tend to produce interferon-gamma: A new player in endometriosis etiology: A case-control study
Abstract
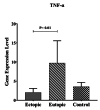
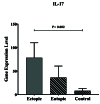
Background: Endometriosis is a chronic estrogen-related inflammatory disorder that is known by proliferating endometrial cells in a place outside the uterus. The high presence of immune cells in the peritoneal fluid of women with endometriosis confirms the involvement of the immune system in the pathogenesis of the disease. Mucosal-associated invariant T (MAIT) cells play an undeniable impact on mucosal immunity by the production of interleukin-17, interferon-gamma (IFN-γ), and tumor necrosis factor-alpha. The function of the cells in the pathogenesis of endometriosis is less investigated.
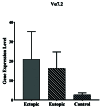
Objective: This study aims to investigate the infiltration of MAIT cells by using the determination levels of V 7.2-J 33 gene expression in eutopic and ectopic tissue of endometriosis lesions.
Materials and methods: In this case-control study, the tested samples include 20 eutopic and 20 ectopic tissues of women with endometriosis and 20 uterine endometrial tissues of women in the control group. Expressions of the V 7.2-J 33 tumor necrosis factor-alpha, interleukin-17A, and IFN-γ genes were analyzed by quantitative reverse transcriptase-polymerase chain reaction.
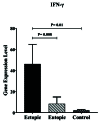
Results: According to the results, V 7.2-J 33 gene expression did not show substantial elevation in the uterine and eutopic endometrial tissues compared to internal gene control as well as in ectopic tissues. Correlation analysis approved a positive relationship between V 7.2-J 33 expression genes and IFN-γ levels in ectopic tissues.
Conclusion: Considering the low-expression specific gene of MAIT cells in ectopic tissue, it can be concluded that these cells are present in the endometriotic environment to a certain extent, and there is a possibility of their role in the progression of endometriosis by secreting IFN- .
Keywords: Endometriosis, MAIT, IFN-γ, TNF-α; IL-17.; TCR V alpha 7.2-J alpha33.
Copyright © 2024 Zare Moghaddam et al.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Figures
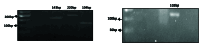
References
-
- Koninckx PR, Ussia A, Adamyan L, Wattiez A, Gomel V, Martin DC. Pathogenesis of endometriosis: The genetic/epigenetic theory. Fertil Steril. 2019;111:327–340. - PubMed
-
- Parazzini F, Esposito G, Tozzi L, Noli S, Bianchi S. Epidemiology of endometriosis and its comorbidities. Eur J Obstet Gynecol Reprod Biol. 2017;209:3–7. - PubMed
-
- Zare Moghaddam M, Ansariniya H, Seifati SM, Zare F, Fesahat F. Immunopathogenesis of endometriosis: An overview of the role of innate and adaptive immune cells and their mediators. Am J Reprod Immunol. 2022;87:e13537. - PubMed
-
- Classen J, Koschel J, Oehlwein C, Seppi K, Urban P, Winkler C, et al. Nonmotor fluctuations: Phenotypes, pathophysiology, management, and open issues. J Neural Transm. 2017;124:1029–1036. - PubMed
LinkOut - more resources
Full Text Sources
