Lenvatinib plus drug-eluting bead transarterial chemoembolization with/without hepatic arterial infusion chemotherapy for hepatocellular carcinoma larger than 7 cm with major portal vein tumor thrombosis: a multicenter retrospective cohort study
- PMID: 38869974
- PMCID: PMC11634077
- DOI: 10.1097/JS9.0000000000001819
Lenvatinib plus drug-eluting bead transarterial chemoembolization with/without hepatic arterial infusion chemotherapy for hepatocellular carcinoma larger than 7 cm with major portal vein tumor thrombosis: a multicenter retrospective cohort study
Abstract
Background: The management of hepatocellular carcinoma (HCC) with high tumor burden and major portal vein tumor thrombosis (PVTT) remains a great challenge. The authors aimed to investigate the efficacy and safety of lenvatinib plus drug-eluting bead transarterial chemoembolization (DEB-TACE) and hepatic arterial infusion chemotherapy (HAIC) with oxaliplatin, fluorouracil and leucovorin (Len+DEB-TACE+HAIC) versus lenvatinib plus DEB-TACE (Len+DEB-TACE) for HCC greater than 7.0 cm accompanied with major PVTT.
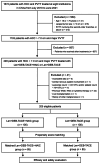
Materials and methods: This multicenter retrospective cohort study evaluated consecutive patients with HCC (> 7.0 cm) and major PVTT who received Len+DEB-TACE+HAIC (Len+DEB-TACE+HAIC group) or Len+DEB-TACE (Len+DEB-TACE group) between July 2019 and June 2021 from eight institutions in China. Objective response rate (ORR), time to progression (TTP), overall survival (OS), and treatment-related adverse events (TRAEs) were compared between the two groups by propensity score matching (PSM).
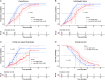
Results: A total of 205 patients were included. After PSM, 85-paired patients remained in the study cohorts. Patients in the Len+DEB-TACE+HAIC group had higher ORR (61.2% vs. 34.1%, P < 0.001), longer TTP (median, 9.8 vs. 5.9 months, P < 0.001), and prolonged OS (median, 16.7 vs. 12.5 months, P < 0.001) than those in the Len+DEB-TACE group. The ORR and TTP of both intrahepatic tumor (ORR: 64.7% vs. 36.5%, P < 0.001; median TTP: 10.7 vs. 7.0 months, P < 0.001) and PVTT (ORR: 74.1% vs. 47.1%, P < 0.001; median TTP: 17.4 vs. 7.6 months, P < 0.001) were better in the Len+DEB-TACE+HAIC group than the Len+DEB-TACE group. The frequency of grade 3-4 TRAEs in the Len+DEB-TACE+HAIC group were comparable to those in the Len+DEB-TACE group (38.8% vs. 34.1%, P = 0.524).
Conclusion: The addition of HAIC to Len+DEB-TACE significantly improved ORR, TTP, and OS over Len+DEB-TACE with an acceptable safety profile for large HCC with major PVTT.
Trial registration: ClinicalTrials.gov NCT06265883.
Copyright © 2024 The Author(s). Published by Wolters Kluwer Health, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article.
Figures

References
-
- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2021;71:209–249. - PubMed
-
- Lu J, Zhang XP, Zhong BY, et al. Management of patients with hepatocellular carcinoma and portal vein tumour thrombosis: comparing east and west. Lancet Gastroenterol Hepatol 2019;4:721–730. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

