A cytokinetic ring-driven cell rotation achieves Hertwig's rule in early development
- PMID: 38870057
- PMCID: PMC11194556
- DOI: 10.1073/pnas.2318838121
A cytokinetic ring-driven cell rotation achieves Hertwig's rule in early development
Abstract
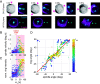
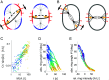
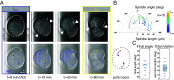
Hertwig's rule states that cells divide along their longest axis, usually driven by forces acting on the mitotic spindle. Here, we show that in contrast to this rule, microtubule-based pulling forces in early Caenorhabditis elegans embryos align the spindle with the short axis of the cell. We combine theory with experiments to reveal that in order to correct this misalignment, inward forces generated by the constricting cytokinetic ring rotate the entire cell until the spindle is aligned with the cell's long axis. Experiments with slightly compressed mouse zygotes indicate that this cytokinetic ring-driven mechanism of ensuring Hertwig's rule is general for cells capable of rotating inside a confining shell, a scenario that applies to early cell divisions of many systems.
Keywords: actomyosin; biophysics; cell biology; cytokinesis; development.
Conflict of interest statement
Competing interests statement:The authors declare no competing interest.
Figures


Similar articles
-
Choice between 1- and 2-furrow cytokinesis in Caenorhabditis elegans embryos with tripolar spindles.Mol Biol Cell. 2019 Jul 22;30(16):2065-2075. doi: 10.1091/mbc.E19-01-0075. Epub 2019 Feb 20. Mol Biol Cell. 2019. PMID: 30785847 Free PMC article.
-
Spindle positioning during the asymmetric first cell division of Caenorhabditis elegans embryos.Novartis Found Symp. 2001;237:164-75; discussion 176-81. doi: 10.1002/0470846666.ch13. Novartis Found Symp. 2001. PMID: 11444042 Review.
-
Physical Limits on the Precision of Mitotic Spindle Positioning by Microtubule Pushing forces: Mechanics of mitotic spindle positioning.Bioessays. 2017 Nov;39(11):10.1002/bies.201700122. doi: 10.1002/bies.201700122. Epub 2017 Sep 28. Bioessays. 2017. PMID: 28960439 Free PMC article. Review.
-
Coupling of rotational cortical flow, asymmetric midbody positioning, and spindle rotation mediates dorsoventral axis formation in C. elegans.Dev Cell. 2014 Feb 10;28(3):253-67. doi: 10.1016/j.devcel.2014.01.002. Dev Cell. 2014. PMID: 24525186
-
Cortical centralspindlin and G alpha have parallel roles in furrow initiation in early C. elegans embryos.J Cell Sci. 2007 May 15;120(Pt 10):1772-8. doi: 10.1242/jcs.03447. Epub 2007 Apr 24. J Cell Sci. 2007. PMID: 17456550
References
-
- Hertwig O., Welchen Einfluss übt die Schwerkraft auf die Theilung der Zellen? (Verlag von Gustav Fischer, Jena, 1884).
-
- Hertwig O., Ueber den Werth der ersten Furchungszellen für die Organbildung des Embryo Experimentelle Studien am Frosch-und Tritonei. Arch. Mikrosk. Anat. 42, 662–807 (1893).
-
- Sachs J., Uber die Anordnung der Zellen in jungsten Pflanzentheilen. Arb. Bot. Inst. Wurzbg. 2, 46–104 (1878).
-
- Wilson E. B., The Cell in Development and Heredity (The Macmillan Company, 1925), (May 24, 2023).
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

