A multi-organ, feto-maternal interface organ-on-chip, models pregnancy pathology and is a useful preclinical extracellular vesicle drug trial platform
- PMID: 38872854
- PMCID: PMC11175617
- DOI: 10.1016/j.vesic.2024.100035
A multi-organ, feto-maternal interface organ-on-chip, models pregnancy pathology and is a useful preclinical extracellular vesicle drug trial platform
Abstract
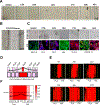
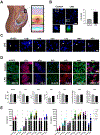
Pregnant women and their fetuses are often excluded from clinical trials due to missing drug-related pre-clinical trial information at the human feto-maternal interface (FMi). The two interfaces-placenta/decidua and fetal membranes/decidua are gatekeepers of drug transport; however, testing their functions is impractical during pregnancy. Limitations of current in-vivo/in-vitro models have hampered drug development and testing during pregnancy. Hence, major complications like preterm births and maternal and neonatal mortalities remain high. Advancements in organ-on-chip (OOC) platforms to test drug kinetics and efficacy and novel extracellular vesicle-based fetal drug delivery are expected to accelerate preclinical trials related to pregnancy complications. Here we report the development and testing of a humanized multi-organ fetal membrane/placenta (fetal)-decidua (maternal) interface OOC (FMi-PLA-OOC) that contains seven cell types interconnected through microchannels to maintain intercellular interactions as seen in-utero. Cytotoxicity, propagation, mechanism of action, and efficacy of engineered extracellular vesicles containing anti-inflammatory interleukin (IL)-10 (eIL-10) were evaluated to reduce FMi inflammation associated with preterm birth. A healthy and disease model (lipopolysaccharide-infectious inflammation) of the FMi-PLA-OOC was created and co-treated with eIL-10. eIL-10 propagated from the maternal to fetal side within 72-hours, localized in all cell types, showed no cytotoxicity, activated IL-10 signaling pathways, and reduced lipopolysaccharide-induced inflammation (minimized NF-kB activation and proinflammatory cytokine production). These data recapitulated eIL-10s' ability to reduce inflammation and delay infection-associated preterm birth in mouse models, suggesting FMi-PLA-OOC as an alternative approach to using animal models. Additionally, we report the utility of eIL-10 that can traverse through FMis to reduce inflammation-associated pregnancy complications.
Keywords: EVs; Fetal membrane; Interleukin-10; Microphysiological systems; Placenta; preterm birth.
Conflict of interest statement
Statements and Declarations: The authors declare no conflicts of interest.
Figures



Similar articles
-
Testing of drugs using human feto-maternal interface organ-on-chips provide insights into pharmacokinetics and efficacy.Lab Chip. 2022 Nov 22;22(23):4574-4592. doi: 10.1039/d2lc00691j. Lab Chip. 2022. PMID: 36322152 Free PMC article.
-
Modeling ascending infection with a feto-maternal interface organ-on-chip.Lab Chip. 2020 Nov 24;20(23):4486-4501. doi: 10.1039/d0lc00875c. Lab Chip. 2020. PMID: 33112317 Free PMC article.
-
Microfluidic technology and simulation models in studying pharmacokinetics during pregnancy.Front Pharmacol. 2023 Aug 17;14:1241815. doi: 10.3389/fphar.2023.1241815. eCollection 2023. Front Pharmacol. 2023. PMID: 37663251 Free PMC article.
-
Organ-On-Chip Technology: The Future of Feto-Maternal Interface Research?Front Physiol. 2020 Jun 30;11:715. doi: 10.3389/fphys.2020.00715. eCollection 2020. Front Physiol. 2020. PMID: 32695021 Free PMC article. Review.
-
Spontaneous preterm birth: Involvement of multiple feto-maternal tissues and organ systems, differing mechanisms, and pathways.Front Endocrinol (Lausanne). 2022 Oct 13;13:1015622. doi: 10.3389/fendo.2022.1015622. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36313741 Free PMC article. Review.
Cited by
-
Exosomal Delivery of Interleukin-10 Reduces Infection-Associated Inflammation in a 3D-Printed Model of a Humanized Feto-Maternal Interface.FASEB J. 2025 May 31;39(10):e70634. doi: 10.1096/fj.202500545R. FASEB J. 2025. PMID: 40356417
-
New approach methods on the bench side to accelerate clinical trials during pregnancy.Expert Opin Drug Metab Toxicol. 2024 Jul;20(7):555-560. doi: 10.1080/17425255.2024.2353944. Epub 2024 May 13. Expert Opin Drug Metab Toxicol. 2024. PMID: 38739076 Free PMC article. Review.
-
Modeling reproductive and pregnancy-associated tissues using organ-on-chip platforms: challenges, limitations, and the high throughput data frontier.Front Bioeng Biotechnol. 2025 Apr 1;13:1568389. doi: 10.3389/fbioe.2025.1568389. eCollection 2025. Front Bioeng Biotechnol. 2025. PMID: 40236940 Free PMC article. Review.
-
Group B streptococcal infections in pregnancy and early life.Clin Microbiol Rev. 2025 Mar 13;38(1):e0015422. doi: 10.1128/cmr.00154-22. Epub 2024 Nov 25. Clin Microbiol Rev. 2025. PMID: 39584819 Review.
-
Lead exposure at the feto-maternal interface: a cause for concern for fetal membrane trophoblasts.Toxicol Sci. 2025 Feb 1;203(2):195-205. doi: 10.1093/toxsci/kfae149. Toxicol Sci. 2025. PMID: 39579145
References
Grants and funding
LinkOut - more resources
Full Text Sources
