A Hydrazone Ligand for Iridium-Catalyzed C-H Borylation: Enhanced Reactivity and Selectivity for Fluorinated Arenes
- PMID: 38873573
- PMCID: PMC11167643
- DOI: 10.1021/acs.organomet.4c00174
A Hydrazone Ligand for Iridium-Catalyzed C-H Borylation: Enhanced Reactivity and Selectivity for Fluorinated Arenes
Abstract
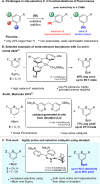
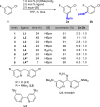
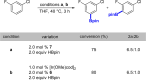
Ir-catalyzed C-H borylations of fluorinated and cyanated arenes with high meta-to-F/CN are described. Use of a dipyridyl hydrazone framework as the ancillary ligand and pinacolborane (HBpin) as the functionalizing reagent generates catalysts that are significantly more active and selective than 4,4'-di-tert-butyl-2,2'-bipyridine (dtbpy) for both electron-deficient and electron-rich substrates. Investigation of the ligand framework resulted in the observation of formal N-borylation of the hydrazone by HBpin, as evidenced by NMR spectroscopy and X-ray crystallography. Subsequent stoichiometric reactions of this adduct with an iridium precatalyst revealed the formation of an unusual IrI hydrazido. Isolation and use of this hydrazido reproduce the selectivity of in situ generated catalysts, suggesting that it leads to formation of the active species.
© 2024 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare the following competing financial interest(s): S.L.M., M.R.S., and R.E.M. own a percentage of BoroPharm, Inc.
Figures

References
LinkOut - more resources
Full Text Sources
