LTβR-RelB signaling in intestinal epithelial cells protects from chemotherapy-induced mucosal damage
- PMID: 38873613
- PMCID: PMC11169669
- DOI: 10.3389/fimmu.2024.1388496
LTβR-RelB signaling in intestinal epithelial cells protects from chemotherapy-induced mucosal damage
Abstract
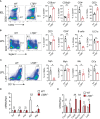
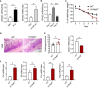
The intricate immune mechanisms governing mucosal healing following intestinal damage induced by cytotoxic drugs remain poorly understood. The goal of this study was to investigate the role of lymphotoxin beta receptor (LTβR) signaling in chemotherapy-induced intestinal damage. LTβR deficient mice exhibited heightened body weight loss, exacerbated intestinal pathology, increased proinflammatory cytokine expression, reduced IL-22 expression, and proliferation of intestinal epithelial cells following methotrexate (MTX) treatment. Furthermore, LTβR-/-IL-22-/- mice succumbed to MTX treatment, suggesting that LTβR- and IL-22- dependent pathways jointly promote mucosal repair. Although both LTβR ligands LIGHT and LTβ were upregulated in the intestine early after MTX treatment, LIGHT-/- mice, but not LTβ-/- mice, displayed exacerbated disease. Further, we revealed the critical role of T cells in mucosal repair as T cell-deficient mice failed to upregulate intestinal LIGHT expression and exhibited increased body weight loss and intestinal pathology. Analysis of mice with conditional inactivation of LTβR revealed that LTβR signaling in intestinal epithelial cells, but not in Lgr5+ intestinal stem cells, macrophages or dendritic cells was critical for mucosal repair. Furthermore, inactivation of the non-canonical NF-kB pathway member RelB in intestinal epithelial cells promoted MTX-induced disease. Based on these results, we propose a model wherein LIGHT produced by T cells activates LTβR-RelB signaling in intestinal epithelial cells to facilitate mucosal repair following chemotherapy treatment.
Keywords: IL-22; LIGHT; LTβR; RelB; intestinal damage; lymphotoxin; methotrexate.
Copyright © 2024 Chen, Muñoz, Korchagina, Shou, Vallecer, Todd, Shein, Tumanov and Koroleva.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision
Figures





Similar articles
-
Lymphotoxin beta receptor signaling limits mucosal damage through driving IL-23 production by epithelial cells.Mucosal Immunol. 2015 Mar;8(2):403-13. doi: 10.1038/mi.2014.78. Epub 2014 Sep 3. Mucosal Immunol. 2015. PMID: 25183367 Free PMC article.
-
Lymphotoxin-β receptor-NIK signaling induces alternative RELB/NF-κB2 activation to promote metastatic gene expression and cell migration in head and neck cancer.Mol Carcinog. 2019 Mar;58(3):411-425. doi: 10.1002/mc.22938. Epub 2018 Nov 28. Mol Carcinog. 2019. PMID: 30488488 Free PMC article.
-
Lymphotoxin β receptor signalling executes Helicobacter pylori-driven gastric inflammation in a T4SS-dependent manner.Gut. 2017 Aug;66(8):1369-1381. doi: 10.1136/gutjnl-2015-310783. Epub 2016 Apr 13. Gut. 2017. PMID: 27196595
-
LTβR Signaling Controls Lymphatic Migration of Immune Cells.Cells. 2021 Mar 29;10(4):747. doi: 10.3390/cells10040747. Cells. 2021. PMID: 33805271 Free PMC article. Review.
-
The LT beta R signaling pathway.Adv Exp Med Biol. 2007;597:160-72. doi: 10.1007/978-0-387-70630-6_13. Adv Exp Med Biol. 2007. PMID: 17633025 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

