Zwitterion-Lubricated Hydrogel Microspheres Encapsulated with Metformin Ameliorate Age-Associated Osteoarthritis
- PMID: 38874373
- PMCID: PMC11321630
- DOI: 10.1002/advs.202402477
Zwitterion-Lubricated Hydrogel Microspheres Encapsulated with Metformin Ameliorate Age-Associated Osteoarthritis
Abstract
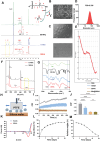
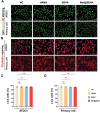
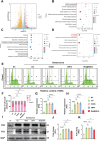
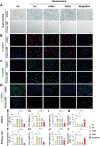
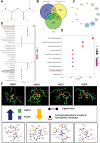
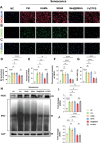
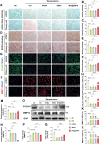
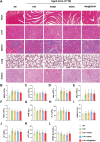
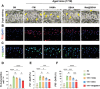
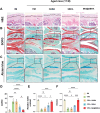
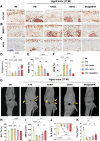
Chondrocyte senescence and reduced lubrication play pivotal roles in the pathogenesis of age-related osteoarthritis (OA). In the present study, highly lubricated and drug-loaded hydrogel microspheres are designed and fabricated through the radical polymerization of sulfobetaine (SB)-modified hyaluronic acid methacrylate using microfluidic technology. The copolymer contains a large number of SB and carboxyl groups that can provide a high degree of lubrication through hydration and form electrostatic loading interactions with metformin (Met@SBHA), producing a high drug load for anti-chondrocyte senescence. Mechanical, tribological, and drug release analyses demonstrated enhanced lubricative properties and prolonged drug dissemination of the Met@SBHA microspheres. RNA sequencing (RNA-seq) analysis, network pharmacology, and in vitro assays revealed the extraordinary capacity of Met@SBHA to combat chondrocyte senescence. Additionally, inducible nitric oxide synthase (iNOS) has been identified as a promising protein modulated by Met in senescent chondrocytes, thereby exerting a significant influence on the iNOS/ONOO-/P53 pathway. Notably, the intra-articular administration of Met@SBHA in aged mice ameliorated cartilage senescence and OA pathogenesis. Based on the findings of this study, Met@SBHA emerges as an innovative and promising strategy in tackling age-related OA serving the dual function of enhancing joint lubrication and mitigating cartilage senescence.
Keywords: age‐related osteoarthritis; cellular senescence; iNOS; lubrication; metformin.
© 2024 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Nicotinamide Adenine Dinucleotide-Loaded Lubricated Hydrogel Microspheres with a Three-Pronged Approach Alleviate Age-Related Osteoarthritis.ACS Nano. 2025 May 13;19(18):17606-17626. doi: 10.1021/acsnano.5c01184. Epub 2025 May 2. ACS Nano. 2025. PMID: 40315404 Free PMC article.
-
In Situ Remodeling of Efferocytosis via Lesion-Localized Microspheres to Reverse Cartilage Senescence.Adv Sci (Weinh). 2024 May;11(19):e2400345. doi: 10.1002/advs.202400345. Epub 2024 Mar 13. Adv Sci (Weinh). 2024. PMID: 38477444 Free PMC article.
-
Biomimetic injectable hydrogel microspheres with enhanced lubrication and controllable drug release for the treatment of osteoarthritis.Bioact Mater. 2021 Mar 26;6(10):3596-3607. doi: 10.1016/j.bioactmat.2021.03.022. eCollection 2021 Oct. Bioact Mater. 2021. PMID: 33869900 Free PMC article.
-
Senescence in osteoarthritis: from mechanism to potential treatment.Arthritis Res Ther. 2022 Jul 22;24(1):174. doi: 10.1186/s13075-022-02859-x. Arthritis Res Ther. 2022. PMID: 35869508 Free PMC article. Review.
-
Cellular senescence in osteoarthritis and anti-aging strategies.Mech Ageing Dev. 2018 Oct;175:83-87. doi: 10.1016/j.mad.2018.08.002. Epub 2018 Aug 11. Mech Ageing Dev. 2018. PMID: 30107185 Review.
Cited by
-
Zwitterionic poly-carboxybetaine-dexamethasone conjugates do not alleviate cartilage degeneration and synovitis in the collagenase-induced osteoarthritis model in rats.Sci Rep. 2025 Jul 1;15(1):20501. doi: 10.1038/s41598-025-93247-3. Sci Rep. 2025. PMID: 40595272 Free PMC article.
-
Injectable microspheres filled with copper-containing bioactive glass improve articular cartilage healing by regulating inflammation and recruiting stem cells.Regen Biomater. 2024 Dec 17;12:rbae142. doi: 10.1093/rb/rbae142. eCollection 2025. Regen Biomater. 2024. PMID: 39845144 Free PMC article.
-
Targeting p21-Positive Senescent Chondrocytes via IL-6R/JAK2 Inhibition to Alleviate Osteoarthritis.Adv Sci (Weinh). 2025 Mar;12(11):e2410795. doi: 10.1002/advs.202410795. Epub 2025 Jan 23. Adv Sci (Weinh). 2025. PMID: 39853717 Free PMC article.
-
A Core-Brush Nanoplatform with Enhanced Lubrication and Anti-Inflammatory Properties for Osteoarthritis Treatment.Adv Sci (Weinh). 2024 Dec;11(47):e2406027. doi: 10.1002/advs.202406027. Epub 2024 Nov 1. Adv Sci (Weinh). 2024. PMID: 39484792 Free PMC article.
-
Preparation of hydrogel microsphere and its application in articular cartilage injury.Mater Today Bio. 2025 Mar 8;31:101641. doi: 10.1016/j.mtbio.2025.101641. eCollection 2025 Apr. Mater Today Bio. 2025. PMID: 40130039 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
- 82272250/National Natural Science Foundation of China
- 81830079/National Natural Science Foundation of China
- 2024A1515012533/Natural Science Foundation of Guangdong Province
- 2021YQPY008/Outstanding Youths Development Scheme of Southern Medical University
- 2022A1515110904/Guangdong Basic and Applied Basic Research Foundation
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
