An integrative sensor of body states: how the mushroom body modulates behavior depending on physiological context
- PMID: 38876486
- PMCID: PMC11199956
- DOI: 10.1101/lm.053918.124
An integrative sensor of body states: how the mushroom body modulates behavior depending on physiological context
Abstract
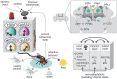
The brain constantly compares past and present experiences to predict the future, thereby enabling instantaneous and future behavioral adjustments. Integration of external information with the animal's current internal needs and behavioral state represents a key challenge of the nervous system. Recent advancements in dissecting the function of the Drosophila mushroom body (MB) at the single-cell level have uncovered its three-layered logic and parallel systems conveying positive and negative values during associative learning. This review explores a lesser-known role of the MB in detecting and integrating body states such as hunger, thirst, and sleep, ultimately modulating motivation and sensory-driven decisions based on the physiological state of the fly. State-dependent signals predominantly affect the activity of modulatory MB input neurons (dopaminergic, serotoninergic, and octopaminergic), but also induce plastic changes directly at the level of the MB intrinsic and output neurons. Thus, the MB emerges as a tightly regulated relay station in the insect brain, orchestrating neuroadaptations due to current internal and behavioral states leading to short- but also long-lasting changes in behavior. While these adaptations are crucial to ensure fitness and survival, recent findings also underscore how circuit motifs in the MB may reflect fundamental design principles that contribute to maladaptive behaviors such as addiction or depression-like symptoms.
© 2024 Suárez-Grimalt et al.; Published by Cold Spring Harbor Laboratory Press.
Figures


References
-
- Agrawal P, Chung P, Heberlein U, Kent C. 2019. Enabling cell-type-specific behavioral epigenetics in Drosophila: a modified high-yield INTACT method reveals the impact of social environment on the epigenetic landscape in dopaminergic neurons. BMC Biol 17: 30. 10.1186/s12915-019-0646-4 - DOI - PMC - PubMed
-
- Araujo SM, Poetini MR, Bortolotto VC, de Freitas Couto S, Pinheiro FC, Meichtry LB, de Almeida FP, Santos Musachio EA, de Paula MT, Prigol M. 2018. Chronic unpredictable mild stress-induced depressive-like behavior and dysregulation of brain levels of biogenic amines in Drosophila melanogaster. Behav Brain Res 351: 104–113. 10.1016/j.bbr.2018.05.016 - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
