Liraglutide exhibits potential anti-tumor effects on the progression of intrahepatic cholangiocarcinoma, in vitro and in vivo
- PMID: 38877189
- PMCID: PMC11178799
- DOI: 10.1038/s41598-024-64774-2
Liraglutide exhibits potential anti-tumor effects on the progression of intrahepatic cholangiocarcinoma, in vitro and in vivo
Abstract
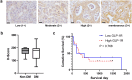
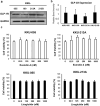
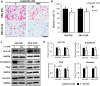
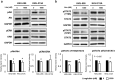
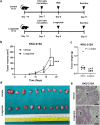
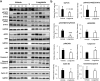
Glucagon-like peptide 1 receptor (GLP-1R) agonist is an emerging anti-diabetic medication whose effects on the risk and progression of cholangiocarcinoma (CCA) are controversial. This study aimed to elucidate the roles of GLP-1R and its agonists on intrahepatic CCA (iCCA) progression. Expressions of GLP-1R in iCCA tissues investigated by immunohistochemistry showed that GLP-1R expressions were significantly associated with poor histological grading (P = 0.027). iCCA cell lines, KKU-055 and KKU-213A, were treated with exendin-4 and liraglutide, GLP-1R agonists, and their effects on proliferation and migration were assessed. Exendin-4 and liraglutide did not affect CCA cell proliferation in vitro, but liraglutide significantly suppressed the migration of CCA cells, partly by inhibiting epithelial-mesenchymal transition. In contrast, liraglutide significantly reduced CCA tumor volumes and weights in xenografted mice (P = 0.046). GLP-1R appeared downregulated when CCA cells were treated with liraglutide in vitro and in vivo. In addition, liraglutide treatment significantly suppressed Akt and STAT3 signaling in CCA cells, by reducing their phosphorylation levels. These results suggested that liraglutide potentially slows down CCA progression, and further clinical investigation would benefit the treatment of CCA with diabetes mellitus.
Keywords: Cholangiocarcinoma; Diabetes mellitus; Glucagon-like peptide 1 receptor; Liraglutide.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Glucagon-like peptide 1 receptor agonist: A potential game changer for cholangiocarcinoma.World J Gastroenterol. 2024 Sep 14;30(34):3862-3867. doi: 10.3748/wjg.v30.i34.3862. World J Gastroenterol. 2024. PMID: 39350782 Free PMC article. Review.
-
Activation of glucagon-like peptide-1 receptor inhibits tumourigenicity and metastasis of human pancreatic cancer cells via PI3K/Akt pathway.Diabetes Obes Metab. 2014 Sep;16(9):850-60. doi: 10.1111/dom.12291. Epub 2014 Apr 15. Diabetes Obes Metab. 2014. PMID: 24641303
-
Allicin Inhibits Proliferation and Invasion in Vitro and in Vivo via SHP-1-Mediated STAT3 Signaling in Cholangiocarcinoma.Cell Physiol Biochem. 2018;47(2):641-653. doi: 10.1159/000490019. Epub 2018 May 22. Cell Physiol Biochem. 2018. PMID: 29794468
-
Increased activation of PI3K/AKT signaling pathway is associated with cholangiocarcinoma metastasis and PI3K/mTOR inhibition presents a possible therapeutic strategy.Tumour Biol. 2013 Dec;34(6):3637-48. doi: 10.1007/s13277-013-0945-2. Epub 2013 Jul 6. Tumour Biol. 2013. PMID: 23832540
-
The cholangiocyte primary cilium in health and disease.Biochim Biophys Acta Mol Basis Dis. 2018 Apr;1864(4 Pt B):1245-1253. doi: 10.1016/j.bbadis.2017.06.006. Epub 2017 Jun 15. Biochim Biophys Acta Mol Basis Dis. 2018. PMID: 28625917 Free PMC article. Review.
Cited by
-
Semaglutide, a glucagon-like peptide-1 receptor agonist, inhibits oral squamous cell carcinoma growth through P38 MAPK signaling pathway.J Cancer Res Clin Oncol. 2025 Mar 7;151(3):103. doi: 10.1007/s00432-025-06154-5. J Cancer Res Clin Oncol. 2025. PMID: 40055197 Free PMC article.
-
Glucagon-like peptide 1 receptor agonist: A potential game changer for cholangiocarcinoma.World J Gastroenterol. 2024 Sep 14;30(34):3862-3867. doi: 10.3748/wjg.v30.i34.3862. World J Gastroenterol. 2024. PMID: 39350782 Free PMC article. Review.
-
The emerging role of GLP-1 receptor agonists in treating or preventing cancer.Cancer Drug Resist. 2024 Dec 7;7:49. doi: 10.20517/cdr.2024.116. eCollection 2024. Cancer Drug Resist. 2024. PMID: 39931650 Free PMC article.
-
Prognostic impact of glucagon-like peptide-1 receptor (GLP1R) expression on cancer survival and its implications for GLP-1R agonist therapy: an integrative analysis across multiple tumor types.Geroscience. 2025 Jun;47(3):4413-4427. doi: 10.1007/s11357-024-01494-5. Epub 2025 Jan 8. Geroscience. 2025. PMID: 39777709 Free PMC article.
-
The Constellation of Risk Factors and Paraneoplastic Syndromes in Cholangiocarcinoma: Integrating the Endocrine Panel Amid Tumour-Related Biology (A Narrative Review).Biology (Basel). 2024 Aug 26;13(9):662. doi: 10.3390/biology13090662. Biology (Basel). 2024. PMID: 39336089 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

