Evaluation of novel Epstein-Barr virus-derived antigen formulations for monitoring virus-specific T cells in pediatric patients with infectious mononucleosis
- PMID: 38877590
- PMCID: PMC11179387
- DOI: 10.1186/s12985-024-02411-0
Evaluation of novel Epstein-Barr virus-derived antigen formulations for monitoring virus-specific T cells in pediatric patients with infectious mononucleosis
Abstract
Background: Infection with the Epstein-Barr virus (EBV) elicits a complex T-cell response against a broad range of viral proteins. Hence, identifying potential differences in the cellular immune response of patients with different EBV-associated diseases or different courses of the same disorder requires interrogation of a maximum number of EBV antigens. Here, we tested three novel EBV-derived antigen formulations for their ability to reactivate virus-specific T cells ex vivo in patients with EBV-associated infectious mononucleosis (IM).
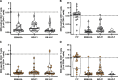
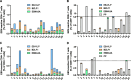
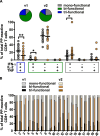
Methods: We comparatively analyzed EBV-specific CD4+ and CD8+ T-cell responses to three EBV-derived antigen formulations in 20 pediatric patients during the early phase of IM: T-activated EBV proteins (BZLF1, EBNA3A) and EBV-like particles (EB-VLP), both able to induce CD4+ and CD8+ T-cell responses ex vivo, as well as an EBV-derived peptide pool (PP) covering 94 well-characterized CD8+ T-cell epitopes. We assessed the specificity, magnitude, kinetics, and functional characteristics of EBV-specific immune responses at two sequential time points (v1 and v2) within the first six weeks after IM symptom onset (Tonset).
Results: All three tested EBV-derived antigen formulations enabled the detection of EBV-reactive T cells during the early phase of IM without prior T-cell expansion in vitro. EBV-reactive CD4+ and CD8+ T cells were mainly mono-functional (CD4+: mean 64.92%, range 56.15-71.71%; CD8+: mean 58.55%, range 11.79-85.22%) within the first two weeks after symptom onset (v1) with IFN-γ and TNF-secreting cells representing the majority of mono-functional EBV-reactive T cells. By contrast, PP-reactive CD8+ T cells were primarily bi-functional (>60% at v1 and v2), produced IFN-γ and TNF and had more tri-functional than mono-functional components. We observed a moderate correlation between viral load and EBNA3A, EB-VLP, and PP-reactive CD8+ T cells (rs = 0.345, 0.418, and 0.356, respectively) within the first two weeks after Tonset, but no correlation with the number of detectable EBV-reactive CD4+ T cells.
Conclusions: All three EBV-derived antigen formulations represent innovative and generic recall antigens suitable for monitoring EBV-specific T-cell responses ex vivo. Their combined use facilitates a thorough analysis of EBV-specific T-cell immunity and allows the identification of functional T-cell signatures linked to disease development and severity.
Keywords: Epstein-Barr virus; Immune monitoring; Infectious mononucleosis; Pediatric patients; T-cell response.
© 2024. The Author(s).
Conflict of interest statement
UP received personal fees from Abbott, Abbvie, Arbutus, Gilead, GSK, J & J, MSD, Roche, Sanofi, Sobi, and Vaccitech. UP is a cofounder and share-holder of SCG Cell Therapy. The lab of UP received grants from SCG Cell Therapy. The remaining authors declare no competing interests.
Figures

References
-
- Longnecker RM, Kieff E, Cohen JI. Epstein-Barr virus. In: Knipe DM, Howley PM, editors. Fields Virology. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2013. pp. 1898–1959.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

