Neonatal Chlamydia muridarum respiratory infection causes neuroinflammation within the brainstem during the early postnatal period
- PMID: 38879567
- PMCID: PMC11179230
- DOI: 10.1186/s12974-024-03150-3
Neonatal Chlamydia muridarum respiratory infection causes neuroinflammation within the brainstem during the early postnatal period
Abstract
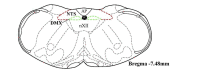
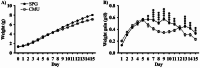
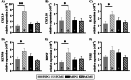
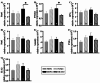
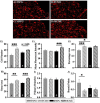
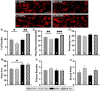
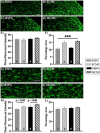
Respiratory infections are one of the most common causes of illness and morbidity in neonates worldwide. In the acute phase infections are known to cause wide-spread peripheral inflammation. However, the inflammatory consequences to the critical neural control centres for respiration have not been explored. Utilising a well characterised model of neonatal respiratory infection, we investigated acute responses within the medulla oblongata which contains key respiratory regions. Neonatal mice were intranasally inoculated within 24 h of birth, with either Chlamydia muridarum or sham-infected, and tissue collected on postnatal day 15, the peak of peripheral inflammation. A key finding of this study is that, while the periphery appeared to show no sex-specific effects of a neonatal respiratory infection, sex had a significant impact on the inflammatory response of the medulla oblongata. There was a distinct sex-specific response in the medulla coincident with peak of peripheral inflammation, with females demonstrating an upregulation of anti-inflammatory cytokines and males showing very few changes. Microglia also demonstrated sex-specificity with the morphology of females and males differing based upon the nuclei. Astrocytes showed limited changes during the acute response to neonatal infection. These data highlight the strong sex-specific impact of a respiratory infection can have on the medulla in the acute inflammatory phase.
Keywords: Astrocytes; Cytokines; Medulla oblongata; Microglia; Sex-specific.
© 2024. The Author(s).
Conflict of interest statement
All authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

Similar articles
-
Characterization of the Horizontal and Vertical Sexual Transmission of Chlamydia Genital Infections in a New Mouse Model.Infect Immun. 2019 Jun 20;87(7):e00834-18. doi: 10.1128/IAI.00834-18. Print 2019 Jul. Infect Immun. 2019. PMID: 30833332 Free PMC article.
-
Chlamydia muridarum plasmid induces mouse oviduct pathology by promoting chlamydial survival and ascending infection and triggering host inflammation.Eur J Dermatol. 2018 Oct 1;28(5):628-636. doi: 10.1684/ejd.2018.3399. Eur J Dermatol. 2018. PMID: 30442635
-
Enhanced virulence of Chlamydia muridarum respiratory infections in the absence of TLR2 activation.PLoS One. 2011;6(6):e20846. doi: 10.1371/journal.pone.0020846. Epub 2011 Jun 14. PLoS One. 2011. PMID: 21695078 Free PMC article.
-
IgA modulates respiratory dysfunction as a sequela to pulmonary chlamydial infection as neonates.Pathog Dis. 2016 Apr;74(3):ftv121. doi: 10.1093/femspd/ftv121. Epub 2016 Jan 10. Pathog Dis. 2016. PMID: 26755533 Free PMC article.
-
Chlamydia Spreading from the Genital Tract to the Gastrointestinal Tract - A Two-Hit Hypothesis.Trends Microbiol. 2018 Jul;26(7):611-623. doi: 10.1016/j.tim.2017.12.002. Epub 2017 Dec 27. Trends Microbiol. 2018. PMID: 29289422 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

