Mass spectrometry imaging of Arabidopsis thaliana with in vivo D2O labeling
- PMID: 38882571
- PMCID: PMC11176549
- DOI: 10.3389/fpls.2024.1379299
Mass spectrometry imaging of Arabidopsis thaliana with in vivo D2O labeling
Abstract
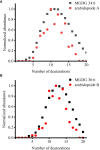
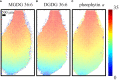
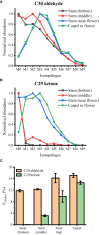
The commonly used analytical tools for metabolomics cannot directly probe metabolic activities or distinguish metabolite differences between cells and suborgans in multicellular organisms. These issues can be addressed by in-vivo isotope labeling and mass spectrometry imaging (MSI), respectively, but the combination of the two, a newly emerging technology we call MSIi, has been rarely applied to plant systems. In this study, we explored MSIi of Arabidopsis thaliana with D2O labeling to study and visualize D-labeling in three classes of lipids: arabidopsides, chloroplast lipids, and epicuticular wax. Similar to other stress responses, D2O-induced stress increased arabidopsides in an hour, but it was relatively minor for matured plants and reverted to the normal level in a few hours. The D-labeling isotopologue patterns of arabidopsides matched with those of galactolipid precursors, supporting the currently accepted biosynthesis mechanism. Matrix-assisted laser desorption/ionization (MALDI)-MSI was used to visualize the spatiotemporal distribution of deuterated chloroplast lipids, pheophytin a, MGDGs, and DGDGs, after growing day-after-sowing (DAS) 28 plants in D2O condition for 3-12 days. There was a gradual change of deuteration amount along the leaf tissues and with a longer labeling time, which was attributed to slow respiration leading to low D2O concentration in the tissues. Finally, deuterium incorporation in epicuticular wax was visualized on the surfaces of the stem and flower. The conversion efficiency of newly synthesized C30 aldehyde to C29 ketone was very low in the lower stem but very high at the top of the stem near the flower or on the flower carpel. This study successfully demonstrated that MSIi can unveil spatiotemporal metabolic activities in various tissues of A. thaliana.
Keywords: Arabidopsis thaliana; arabidopsides; chloroplast lipids; epicuticular wax; in vivo isotope labeling; mass spectrometry imaging; matrix-assisted laser desorption/ionization.
Copyright © 2024 Na and Lee.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Spatio-Temporal Study of Galactolipid Biosynthesis in Duckweed Using Mass Spectrometry Imaging and in vivo Isotope Labeling.Plant Cell Physiol. 2024 Jun 27;65(6):986-998. doi: 10.1093/pcp/pcae032. Plant Cell Physiol. 2024. PMID: 38590126 Free PMC article.
-
FERONIA mutation induces high levels of chloroplast-localized Arabidopsides which are involved in root growth.Plant J. 2019 Jan;97(2):341-351. doi: 10.1111/tpj.14123. Epub 2018 Nov 5. Plant J. 2019. PMID: 30300943
-
Direct profiling and imaging of epicuticular waxes on Arabidopsis thaliana by laser desorption/ionization mass spectrometry using silver colloid as a matrix.Anal Chem. 2009 Apr 15;81(8):2991-3000. doi: 10.1021/ac802615r. Anal Chem. 2009. PMID: 19290666
-
Recent advances in matrix-assisted laser desorption/ionisation mass spectrometry imaging (MALDI-MSI) for in situ analysis of endogenous molecules in plants.Phytochem Anal. 2018 Jul;29(4):351-364. doi: 10.1002/pca.2759. Epub 2018 Apr 17. Phytochem Anal. 2018. PMID: 29667236 Review.
-
Metabolic deuterium oxide (D2O) labeling in quantitative omics studies: A tutorial review.Anal Chim Acta. 2023 Feb 15;1242:340722. doi: 10.1016/j.aca.2022.340722. Epub 2022 Dec 14. Anal Chim Acta. 2023. PMID: 36657897 Review.
Cited by
-
Quantitative ambient mass spectrometry imaging in plants: A perspective on challenges and future applications.Curr Opin Plant Biol. 2025 Jun;85:102736. doi: 10.1016/j.pbi.2025.102736. Epub 2025 May 19. Curr Opin Plant Biol. 2025. PMID: 40393357 Free PMC article. Review.
References
-
- Åstot C., Dolezal K., Moritz T., Sandberg G. (2000). Deuterium in vivo labelling of cytokinins in Arabidopsis thaliana analysed by capillary liquid chromatography/frit-fast atom bombardment mass spectrometry. J. Mass Spectrom 35, 13–22. doi: 10.1002/(SICI)1096-9888(200001)35:1<13::AID-JMS901>3.0.CO;2-I - DOI - PubMed
-
- Buseman C. M., Tamura P., Sparks A. A., Baughman E. J., Maatta S., Zhao J., et al. . (2006). Wounding stimulates the accumulation of glycerolipids containing oxophytodienoic acid and dinor-oxophytodienoic acid in arabidopsis leaves. Plant Physiol. 142, 28–39. doi: 10.1104/pp.106.082115 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

