Maintaining moderate levels of hypochlorous acid promotes neural stem cell proliferation and differentiation in the recovery phase of stroke
- PMID: 38886957
- PMCID: PMC11433893
- DOI: 10.4103/1673-5374.392889
Maintaining moderate levels of hypochlorous acid promotes neural stem cell proliferation and differentiation in the recovery phase of stroke
Abstract
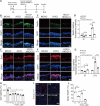
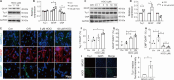
JOURNAL/nrgr/04.03/01300535-202503000-00029/figure1/v/2024-06-17T092413Z/r/image-tiff It has been shown clinically that continuous removal of ischemia/reperfusion-induced reactive oxygen species is not conducive to the recovery of late stroke. Indeed, previous studies have shown that excessive increases in hypochlorous acid after stroke can cause severe damage to brain tissue. Our previous studies have found that a small amount of hypochlorous acid still exists in the later stage of stroke, but its specific role and mechanism are currently unclear. To simulate stroke in vivo, a middle cerebral artery occlusion rat model was established, with an oxygen-glucose deprivation/reoxygenation model established in vitro to mimic stroke. We found that in the early stage (within 24 hours) of ischemic stroke, neutrophils produced a large amount of hypochlorous acid, while in the recovery phase (10 days after stroke), microglia were activated and produced a small amount of hypochlorous acid. Further, in acute stroke in rats, hypochlorous acid production was prevented using a hypochlorous acid scavenger, taurine, or myeloperoxidase inhibitor, 4-aminobenzoic acid hydrazide. Our results showed that high levels of hypochlorous acid (200 μM) induced neuronal apoptosis after oxygen/glucose deprivation/reoxygenation. However, in the recovery phase of the middle cerebral artery occlusion model, a moderate level of hypochlorous acid promoted the proliferation and differentiation of neural stem cells into neurons and astrocytes. This suggests that hypochlorous acid plays different roles at different phases of cerebral ischemia/reperfusion injury. Lower levels of hypochlorous acid (5 and 100 μM) promoted nuclear translocation of β-catenin. By transfection of single-site mutation plasmids, we found that hypochlorous acid induced chlorination of the β-catenin tyrosine 30 residue, which promoted nuclear translocation. Altogether, our study indicates that maintaining low levels of hypochlorous acid plays a key role in the recovery of neurological function.
Copyright © 2025 Copyright: © 2025 Neural Regeneration Research.
Conflict of interest statement
Figures




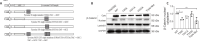
Similar articles
-
Hypochlorous acid derived from microglial myeloperoxidase could mediate high-mobility group box 1 release from neurons to amplify brain damage in cerebral ischemia-reperfusion injury.J Neuroinflammation. 2024 Mar 21;21(1):70. doi: 10.1186/s12974-023-02991-8. J Neuroinflammation. 2024. PMID: 38515139 Free PMC article.
-
High-frequency repetitive transcranial magnetic stimulation promotes neural stem cell proliferation after ischemic stroke.Neural Regen Res. 2024 Aug 1;19(8):1772-1780. doi: 10.4103/1673-5374.389303. Epub 2023 Nov 8. Neural Regen Res. 2024. PMID: 38103244 Free PMC article.
-
Mailuoning oral liquid attenuates convalescent cerebral ischemia by inhibiting AMPK/mTOR-associated apoptosis and promoting CREB/BDNF-mediated neuroprotection.J Ethnopharmacol. 2023 Dec 5;317:116731. doi: 10.1016/j.jep.2023.116731. Epub 2023 Jun 3. J Ethnopharmacol. 2023. PMID: 37277084
-
Effects of primary microglia and astrocytes on neural stem cells in in vitro and in vivo models of ischemic stroke.Neural Regen Res. 2021 Sep;16(9):1677-1685. doi: 10.4103/1673-5374.306093. Neural Regen Res. 2021. PMID: 33510055 Free PMC article.
-
Tanshinone IIA Alleviates Traumatic Brain Injury by Reducing Ischemia‒Reperfusion via the miR-124-5p/FoxO1 Axis.Mediators Inflamm. 2024 Mar 21;2024:7459054. doi: 10.1155/2024/7459054. eCollection 2024. Mediators Inflamm. 2024. PMID: 38549714 Free PMC article.
Cited by
-
A Colorimetric/Ratiometric Fluorescent Probe Based on Aggregation-Induced Emission Effect for Detecting Hypochlorous Acid in Real Samples and Bioimaging Applications.Foods. 2025 Jul 16;14(14):2491. doi: 10.3390/foods14142491. Foods. 2025. PMID: 40724314 Free PMC article.
References
-
- Augsburger F, Filippova A, Jaquet V. Methods for detection of NOX-derived superoxide radical anion and hydrogen peroxide in cells. Methods Mol Biol. 20191982:233–241. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

