Associations of Plasma Glutamatergic Metabolites with Alpha Desynchronization during Cognitive Interference and Working Memory Tasks in Asymptomatic Alzheimer's Disease
- PMID: 38891102
- PMCID: PMC11171970
- DOI: 10.3390/cells13110970
Associations of Plasma Glutamatergic Metabolites with Alpha Desynchronization during Cognitive Interference and Working Memory Tasks in Asymptomatic Alzheimer's Disease
Abstract
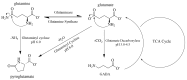
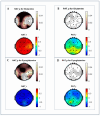
Electroencephalogram (EEG) studies have suggested compensatory brain overactivation in cognitively healthy (CH) older adults with pathological beta-amyloid(Aβ42)/tau ratios during working memory and interference processing. However, the association between glutamatergic metabolites and brain activation proxied by EEG signals has not been thoroughly investigated. We aim to determine the involvement of these metabolites in EEG signaling. We focused on CH older adults classified under (1) normal CSF Aβ42/tau ratios (CH-NATs) and (2) pathological Aβ42/tau ratios (CH-PATs). We measured plasma glutamine, glutamate, pyroglutamate, and γ-aminobutyric acid concentrations using tandem mass spectrometry and conducted a correlational analysis with alpha frequency event-related desynchronization (ERD). Under the N-back working memory paradigm, CH-NATs presented negative correlations (r = ~-0.74--0.96, p = 0.0001-0.0414) between pyroglutamate and alpha ERD but positive correlations (r = ~0.82-0.95, p = 0.0003-0.0119) between glutamine and alpha ERD. Under Stroop interference testing, CH-NATs generated negative correlations between glutamine and left temporal alpha ERD (r = -0.96, p = 0.037 and r = -0.97, p = 0.027). Our study demonstrated that glutamine and pyroglutamate levels were associated with EEG activity only in CH-NATs. These results suggest cognitively healthy adults with amyloid/tau pathology experience subtle metabolic dysfunction that may influence EEG signaling during cognitive challenge. A longitudinal follow-up study with a larger sample size is needed to validate these pilot studies.
Keywords: Alzheimer’s disease (AD); electroencephalogram (EEG); event-related alpha desynchronization (ERD); glutamate; glutamine; pyroglutamate.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the study’s design, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Figures
References
-
- Harrington M.G., Chiang J., Pogoda J.M., Gomez M., Thomas K., Marion S.D., Miller K.J., Siddarth P., Yi X., Zhou F., et al. Executive function changes before memory in preclinical Alzheimer’s pathology: A prospective, cross-sectional, case control study. PLoS ONE. 2013;8:e79378. doi: 10.1371/journal.pone.0079378. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

