Experimental Autoimmune Encephalomyelitis Influences GH-Axis in Female Rats
- PMID: 38892024
- PMCID: PMC11172041
- DOI: 10.3390/ijms25115837
Experimental Autoimmune Encephalomyelitis Influences GH-Axis in Female Rats
Abstract
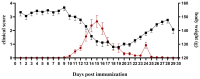
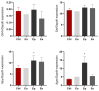
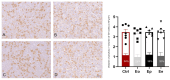
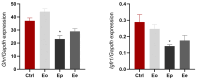
Inflammation, demyelination, and axonal damage to the central nervous system (CNS) are the hallmarks of multiple sclerosis (MS) and its representative animal model, experimental autoimmune encephalomyelitis (EAE). There is scientific evidence for the involvement of growth hormone (GH) in autoimmune regulation. Previous data on the relationship between the GH/insulin like growth factor-1 (IGF-1) axis and MS/EAE are inconclusive; therefore, the aim of our study was to investigate the changes in the GH axis during acute monophasic EAE. The results show that the gene expression of Ghrh and Sst in the hypothalamus does not change, except for Npy and Agrp, while at the pituitary level the Gh, Ghrhr and Ghr genes are upregulated. Interestingly, the cell volume of somatotropic cells in the pituitary gland remains unchanged at the peak of the disease. We found elevated serum GH levels in association with low IGF-1 concentration and downregulated Ghr and Igf1r expression in the liver, indicating a condition resembling GH resistance. This is likely due to inadequate nutrient intake at the peak of the disease when inflammation in the CNS is greatest. Considering that GH secretion is finely regulated by numerous central and peripheral signals, the involvement of the GH/IGF-1 axis in MS/EAE should be thoroughly investigated for possible future therapeutic strategies, especially with a view to improving EAE disease.
Keywords: EAE; IGF-1; growth hormone; pituitary.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
GH-Releasing Hormone Neurons Regulate the Hypothalamic-Pituitary-Somatotropic Axis via Short-Loop Negative Feedback.Endocrinology. 2025 Mar 24;166(5):bqaf062. doi: 10.1210/endocr/bqaf062. Endocrinology. 2025. PMID: 40172534
-
Increase in mRNA concentrations of pituitary receptors for growth hormone-releasing hormone and growth hormone secretagogues after neonatal monosodium glutamate treatment.J Neuroendocrinol. 2000 Apr;12(4):335-41. doi: 10.1046/j.1365-2826.2000.00458.x. J Neuroendocrinol. 2000. PMID: 10718930
-
Pituitary mRNA expression of the growth hormone axis in the 1-year-old intrauterine growth restricted rat.J Neuroendocrinol. 2006 Aug;18(8):611-20. doi: 10.1111/j.1365-2826.2006.01451.x. J Neuroendocrinol. 2006. PMID: 16867182
-
Genetics of GHRH, GHRH-receptor, GH and GH-receptor: its impact on pharmacogenetics.Best Pract Res Clin Endocrinol Metab. 2011 Feb;25(1):25-41. doi: 10.1016/j.beem.2010.06.006. Best Pract Res Clin Endocrinol Metab. 2011. PMID: 21396573 Review.
-
The Complex World of Regulation of Pituitary Growth Hormone Secretion: The Role of Ghrelin, Klotho, and Nesfatins in It.Front Endocrinol (Lausanne). 2021 Mar 11;12:636403. doi: 10.3389/fendo.2021.636403. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 33776931 Free PMC article. Review.
Cited by
-
Special Issue "Advances in Molecular Research on Autoimmune Diseases".Int J Mol Sci. 2024 Oct 25;25(21):11487. doi: 10.3390/ijms252111487. Int J Mol Sci. 2024. PMID: 39519041 Free PMC article.
-
The history of an effective, specific and sensitive diagnostic test: the GHRH test in clinical practice.Rev Endocr Metab Disord. 2025 Jun;26(3):353-369. doi: 10.1007/s11154-024-09938-1. Epub 2024 Dec 17. Rev Endocr Metab Disord. 2025. PMID: 39681762 Review.
-
The Lemon Flavonoid Eriomin® Suppresses Pituitary-Adrenal Axis Activity in Aged Rats.Int J Mol Sci. 2025 Jun 17;26(12):5818. doi: 10.3390/ijms26125818. Int J Mol Sci. 2025. PMID: 40565281 Free PMC article.
-
GHRH in diabetes and metabolism.Rev Endocr Metab Disord. 2025 Jun;26(3):413-426. doi: 10.1007/s11154-024-09930-9. Epub 2024 Nov 19. Rev Endocr Metab Disord. 2025. PMID: 39560873 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

