Equine Endothelial Cells Show Pro-Angiogenic Behaviours in Response to Fibroblast Growth Factor 2 but Not Vascular Endothelial Growth Factor A
- PMID: 38892205
- PMCID: PMC11172845
- DOI: 10.3390/ijms25116017
Equine Endothelial Cells Show Pro-Angiogenic Behaviours in Response to Fibroblast Growth Factor 2 but Not Vascular Endothelial Growth Factor A
Abstract
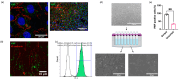
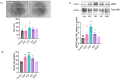
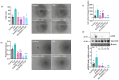
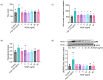
Understanding the factors which control endothelial cell (EC) function and angiogenesis is crucial for developing the horse as a disease model, but equine ECs remain poorly studied. In this study, we have optimised methods for the isolation and culture of equine aortic endothelial cells (EAoECs) and characterised their angiogenic functions in vitro. Mechanical dissociation, followed by magnetic purification using an anti-VE-cadherin antibody, resulted in EC-enriched cultures suitable for further study. Fibroblast growth factor 2 (FGF2) increased the EAoEC proliferation rate and stimulated scratch wound closure and tube formation by EAoECs on the extracellular matrix. Pharmacological inhibitors of FGF receptor 1 (FGFR1) (SU5402) or mitogen-activated protein kinase (MEK) (PD184352) blocked FGF2-induced extracellular signal-regulated kinase 1/2 (ERK1/2) phosphorylation and functional responses, suggesting that these are dependent on FGFR1/MEK-ERK signalling. In marked contrast, vascular endothelial growth factor-A (VEGF-A) had no effect on EAoEC proliferation, migration, or tubulogenesis and did not promote ERK1/2 phosphorylation, indicating a lack of sensitivity to this classical pro-angiogenic growth factor. Gene expression analysis showed that unlike human ECs, FGFR1 is expressed by EAoECs at a much higher level than both VEGF receptor (VEGFR)1 and VEGFR2. These results suggest a predominant role for FGF2 versus VEGF-A in controlling the angiogenic functions of equine ECs. Collectively, our novel data provide a sound basis for studying angiogenic processes in horses and lay the foundations for comparative studies of EC biology in horses versus humans.
Keywords: angiogenesis; comparative; endothelial; equine.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


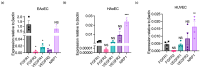
Similar articles
-
Caveolin-1 orchestrates fibroblast growth factor 2 signaling control of angiogenesis in placental artery endothelial cell caveolae.J Cell Physiol. 2012 Jun;227(6):2480-91. doi: 10.1002/jcp.22984. J Cell Physiol. 2012. PMID: 21830216 Free PMC article.
-
L-5F, an apolipoprotein A-I mimetic, inhibits tumor angiogenesis by suppressing VEGF/basic FGF signaling pathways.Integr Biol (Camb). 2011 Apr;3(4):479-89. doi: 10.1039/c0ib00147c. Epub 2011 Feb 1. Integr Biol (Camb). 2011. PMID: 21283904 Free PMC article.
-
In vitro assays using primary embryonic mouse lymphatic endothelial cells uncover key roles for FGFR1 signalling in lymphangiogenesis.PLoS One. 2012;7(7):e40497. doi: 10.1371/journal.pone.0040497. Epub 2012 Jul 6. PLoS One. 2012. PMID: 22792354 Free PMC article.
-
Prostaglandin E2 regulates angiogenesis via activation of fibroblast growth factor receptor-1.J Biol Chem. 2008 Jan 25;283(4):2139-46. doi: 10.1074/jbc.M703090200. Epub 2007 Nov 26. J Biol Chem. 2008. PMID: 18042549
-
Fibroblast growth factor regulation of neovascularization.Curr Opin Hematol. 2008 May;15(3):215-20. doi: 10.1097/MOH.0b013e3282f97d98. Curr Opin Hematol. 2008. PMID: 18391788 Free PMC article. Review.
Cited by
-
New Interpretations for Sprouting, Intussusception, Ansiform, and Coalescent Types of Angiogenesis.Int J Mol Sci. 2024 Aug 6;25(16):8575. doi: 10.3390/ijms25168575. Int J Mol Sci. 2024. PMID: 39201261 Free PMC article. Review.
References
-
- Lawal T.A., Wires E.S., Terry N.L., Dowling J.J., Todd J.J. Preclinical model systems of ryanodine receptor 1-related myopathies and malignant hyperthermia: A comprehensive scoping review of works published 1990–2019. Orphanet J. Rare Dis. 2020;15:113. doi: 10.1186/s13023-020-01384-x. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

