Natural Product-Derived Compounds Targeting Keratinocytes and Molecular Pathways in Psoriasis Therapeutics
- PMID: 38892253
- PMCID: PMC11172960
- DOI: 10.3390/ijms25116068
Natural Product-Derived Compounds Targeting Keratinocytes and Molecular Pathways in Psoriasis Therapeutics
Abstract
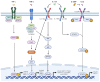
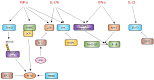
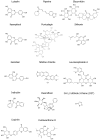
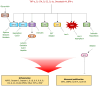
Psoriasis is a chronic autoimmune inflammatory skin disorder that affects approximately 2-3% of the global population due to significant genetic predisposition. It is characterized by an uncontrolled growth and differentiation of keratinocytes, leading to the formation of scaly erythematous plaques. Psoriasis extends beyond dermatological manifestations to impact joints and nails and is often associated with systemic disorders. Although traditional treatments provide relief, their use is limited by potential side effects and the chronic nature of the disease. This review aims to discuss the therapeutic potential of keratinocyte-targeting natural products in psoriasis and highlight their efficacy and safety in comparison with conventional treatments. This review comprehensively examines psoriasis pathogenesis within keratinocytes and the various related signaling pathways (such as JAK-STAT and NF-κB) and cytokines. It presents molecular targets such as high-mobility group box-1 (HMGB1), dual-specificity phosphatase-1 (DUSP1), and the aryl hydrocarbon receptor (AhR) for treating psoriasis. It evaluates the ability of natural compounds such as luteolin, piperine, and glycyrrhizin to modulate psoriasis-related pathways. Finally, it offers insights into alternative and sustainable treatment options with fewer side effects.
Keywords: inflammation; keratinocyte; natural products; psoriasis; signaling pathway.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Anti-Inflammatory and Pro-Differentiating Properties of the Aryl Hydrocarbon Receptor Ligands NPD-0614-13 and NPD-0614-24: Potential Therapeutic Benefits in Psoriasis.Int J Mol Sci. 2021 Jul 13;22(14):7501. doi: 10.3390/ijms22147501. Int J Mol Sci. 2021. PMID: 34299118 Free PMC article.
-
Apoptotic or Antiproliferative Activity of Natural Products against Keratinocytes for the Treatment of Psoriasis.Int J Mol Sci. 2019 May 24;20(10):2558. doi: 10.3390/ijms20102558. Int J Mol Sci. 2019. PMID: 31137673 Free PMC article. Review.
-
Role of Aryl Hydrocarbon Receptor Activation and Autophagy in Psoriasis-Related Inflammation.Int J Mol Sci. 2020 Mar 22;21(6):2195. doi: 10.3390/ijms21062195. Int J Mol Sci. 2020. PMID: 32235789 Free PMC article.
-
Natural Compounds Tapinarof and Galactomyces Ferment Filtrate Downregulate IL-33 Expression via the AHR/IL-37 Axis in Human Keratinocytes.Front Immunol. 2022 May 19;13:745997. doi: 10.3389/fimmu.2022.745997. eCollection 2022. Front Immunol. 2022. PMID: 35663970 Free PMC article.
-
Navigating the landscape of psoriasis therapy: novel targeted pathways and emerging trends.Expert Opin Ther Targets. 2023 Jul-Dec;27(12):1247-1256. doi: 10.1080/14728222.2023.2288273. Epub 2023 Dec 30. Expert Opin Ther Targets. 2023. PMID: 37997278 Review.
Cited by
-
Emerging Novel Therapies for the Treatment of Psoriasis: A Narrative Review.Cureus. 2025 Feb 26;17(2):e79693. doi: 10.7759/cureus.79693. eCollection 2025 Feb. Cureus. 2025. PMID: 40161116 Free PMC article. Review.
-
Glycyrrhizic acid and its carrier-free micellar formulation: Unraveling the potential for enhanced oral prevention of hearing loss.Int J Pharm X. 2025 May 26;9:100340. doi: 10.1016/j.ijpx.2025.100340. eCollection 2025 Jun. Int J Pharm X. 2025. PMID: 40521160 Free PMC article.
-
Trans-p-Coumaryl Alcohol as a Bioactive Compound and Anti-Inflammatory Agent in Wannachawee Recipe for Psoriasis.Pharmaceutics. 2025 Jun 30;17(7):864. doi: 10.3390/pharmaceutics17070864. Pharmaceutics. 2025. PMID: 40733073 Free PMC article.
-
Emerging insights into the role of natural products and miRNAs in psoriasis: from pathophysiology to precision medicine.Naunyn Schmiedebergs Arch Pharmacol. 2025 Mar;398(3):2487-2509. doi: 10.1007/s00210-024-03528-3. Epub 2024 Oct 28. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39466441 Review.
-
Modulation of the KEAP1-NRF2 pathway by Erianin: A novel approach to reduce psoriasiform inflammation and inflammatory signaling.Open Life Sci. 2025 Jul 11;20(1):20251139. doi: 10.1515/biol-2025-1139. eCollection 2025. Open Life Sci. 2025. PMID: 40667480 Free PMC article.
References
-
- Valenzuela F., Flores R. Textbook of Dermatologic Ultrasound. Springer; Berlin/Heidelberg, Germany: 2022. Clinical Overview of Psoriasis and Psoriatic Arthritis; pp. 345–365.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

