hTERT Peptide Fragment GV1001 Prevents the Development of Porphyromonas gingivalis-Induced Periodontal Disease and Systemic Disorders in ApoE-Deficient Mice
- PMID: 38892314
- PMCID: PMC11172542
- DOI: 10.3390/ijms25116126
hTERT Peptide Fragment GV1001 Prevents the Development of Porphyromonas gingivalis-Induced Periodontal Disease and Systemic Disorders in ApoE-Deficient Mice
Abstract
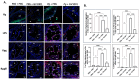
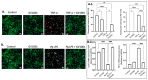
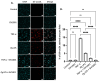
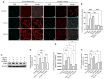
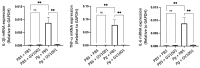
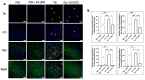
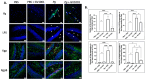
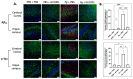
GV1001, an anticancer vaccine, exhibits other biological functions, including anti-inflammatory and antioxidant activity. It also suppresses the development of ligature-induced periodontitis in mice. Porphyromonas gingivalis (Pg), a major human oral bacterium implicated in the development of periodontitis, is associated with various systemic disorders, such as atherosclerosis and Alzheimer's disease (AD). This study aimed to explore the protective effects of GV1001 against Pg-induced periodontal disease, atherosclerosis, and AD-like conditions in Apolipoprotein (ApoE)-deficient mice. GV1001 effectively mitigated the development of Pg-induced periodontal disease, atherosclerosis, and AD-like conditions by counteracting Pg-induced local and systemic inflammation, partly by inhibiting the accumulation of Pg DNA aggregates, Pg lipopolysaccharides (LPS), and gingipains in the gingival tissue, arterial wall, and brain. GV1001 attenuated the development of atherosclerosis by inhibiting vascular inflammation, lipid deposition in the arterial wall, endothelial to mesenchymal cell transition (EndMT), the expression of Cluster of Differentiation 47 (CD47) from arterial smooth muscle cells, and the formation of foam cells in mice with Pg-induced periodontal disease. GV1001 also suppressed the accumulation of AD biomarkers in the brains of mice with periodontal disease. Overall, these findings suggest that GV1001 holds promise as a preventive agent in the development of atherosclerosis and AD-like conditions associated with periodontal disease.
Keywords: Alzheimer disease; GV1001; Porphyromonas gingivalis; atherosclerosis; periodontitis.
Conflict of interest statement
Sangjae Kim is also an employee of GemVax/Kael, Inc., as a scientific consultant, which provided GV1001 to us for this study. The authors declare that this research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest. The funders (UCLA, NIH, and various donors) had no role in the design of the study; in the collection, analysis, or interpretation of data; in the writing of manuscript; or in the decision to publish results. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
GV1001, hTERT Peptide Fragment, Prevents Doxorubicin-Induced Endothelial-to-Mesenchymal Transition in Human Endothelial Cells and Atherosclerosis in Mice.Cells. 2025 Jan 10;14(2):98. doi: 10.3390/cells14020098. Cells. 2025. PMID: 39851526 Free PMC article.
-
GV1001 Inhibits the Severity of the Ligature-Induced Periodontitis and the Vascular Lipid Deposition Associated with the Periodontitis in Mice.Int J Mol Sci. 2023 Aug 8;24(16):12566. doi: 10.3390/ijms241612566. Int J Mol Sci. 2023. PMID: 37628753 Free PMC article.
-
Porphyromonas gingivalis mediated periodontal disease and atherosclerosis: disparate diseases with commonalities in pathogenesis through TLRs.Curr Pharm Des. 2007;13(36):3665-75. doi: 10.2174/138161207783018554. Curr Pharm Des. 2007. PMID: 18220804 Review.
-
Porphyromonas gingivalis infection accelerates the progression of atherosclerosis in a heterozygous apolipoprotein E-deficient murine model.Circulation. 2002 Feb 19;105(7):861-7. doi: 10.1161/hc0702.104178. Circulation. 2002. PMID: 11854128
-
Intranasal immunization with Porphyromonas gingivalis and atherosclerosis.Immunopharmacol Immunotoxicol. 2009;31(3):352-7. doi: 10.1080/08923970902798140. Immunopharmacol Immunotoxicol. 2009. PMID: 19555215 Review.
Cited by
-
GV1001, hTERT Peptide Fragment, Prevents Doxorubicin-Induced Endothelial-to-Mesenchymal Transition in Human Endothelial Cells and Atherosclerosis in Mice.Cells. 2025 Jan 10;14(2):98. doi: 10.3390/cells14020098. Cells. 2025. PMID: 39851526 Free PMC article.
-
Murine Models in Oral Research: A Narrative Review of Experimental Approaches and Cardiovascular Implications.Biology (Basel). 2025 Jan 26;14(2):127. doi: 10.3390/biology14020127. Biology (Basel). 2025. PMID: 40001895 Free PMC article. Review.
References
-
- Kim S., Kim B.J., Kim I., Kim J.H., Kim H.K., Ryu H., Choi D.R., Hwang I.G., Song H., Kwon J.H., et al. A phase II study of chemotherapy in combination with telomerase peptide vaccine (GV1001) as second-line treatment in patients with metastatic colorectal cancer. J. Cancer. 2022;13:1363–1369. doi: 10.7150/jca.70385. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

