Modified mRNA-Mediated CCN5 Gene Transfer Ameliorates Cardiac Dysfunction and Fibrosis without Adverse Structural Remodeling
- PMID: 38892449
- PMCID: PMC11172546
- DOI: 10.3390/ijms25116262
Modified mRNA-Mediated CCN5 Gene Transfer Ameliorates Cardiac Dysfunction and Fibrosis without Adverse Structural Remodeling
Abstract
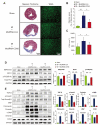
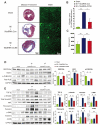
Modified mRNAs (modRNAs) are an emerging delivery method for gene therapy. The success of modRNA-based COVID-19 vaccines has demonstrated that modRNA is a safe and effective therapeutic tool. Moreover, modRNA has the potential to treat various human diseases, including cardiac dysfunction. Acute myocardial infarction (MI) is a major cardiac disorder that currently lacks curative treatment options, and MI is commonly accompanied by fibrosis and impaired cardiac function. Our group previously demonstrated that the matricellular protein CCN5 inhibits cardiac fibrosis (CF) and mitigates cardiac dysfunction. However, it remains unclear whether early intervention of CF under stress conditions is beneficial or more detrimental due to potential adverse effects such as left ventricular (LV) rupture. We hypothesized that CCN5 would alleviate the adverse effects of myocardial infarction (MI) through its anti-fibrotic properties under stress conditions. To induce the rapid expression of CCN5, ModRNA-CCN5 was synthesized and administrated directly into the myocardium in a mouse MI model. To evaluate CCN5 activity, we established two independent experimental schemes: (1) preventive intervention and (2) therapeutic intervention. Functional analyses, including echocardiography and magnetic resonance imaging (MRI), along with molecular assays, demonstrated that modRNA-mediated CCN5 gene transfer significantly attenuated cardiac fibrosis and improved cardiac function in both preventive and therapeutic models, without causing left ventricular rupture or any adverse cardiac remodeling. In conclusion, early intervention in CF by ModRNA-CCN5 gene transfer is an efficient and safe therapeutic modality for treating MI-induced heart failure.
Keywords: CCN5; cardiac fibrosis; heart failure (HF); left ventricular rupture; modified mRNA (modRNA); myocardial infarction (MI).
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose.
Figures


Similar articles
-
Matricellular Protein CCN5 Reverses Established Cardiac Fibrosis.J Am Coll Cardiol. 2016 Apr 5;67(13):1556-1568. doi: 10.1016/j.jacc.2016.01.030. J Am Coll Cardiol. 2016. PMID: 27150688 Free PMC article.
-
Exercise attenuates inflammation and limits scar thinning after myocardial infarction in mice.Am J Physiol Heart Circ Physiol. 2015 Jul 15;309(2):H345-59. doi: 10.1152/ajpheart.00683.2014. Epub 2015 May 22. Am J Physiol Heart Circ Physiol. 2015. PMID: 26001415
-
Cardiac-selective expression of extracellular superoxide dismutase after systemic injection of adeno-associated virus 9 protects the heart against post-myocardial infarction left ventricular remodeling.Circ Cardiovasc Imaging. 2013 May 1;6(3):478-86. doi: 10.1161/CIRCIMAGING.112.000320. Epub 2013 Mar 27. Circ Cardiovasc Imaging. 2013. PMID: 23536266 Free PMC article.
-
Modified mRNA as a Treatment for Myocardial Infarction.Int J Mol Sci. 2023 Mar 1;24(5):4737. doi: 10.3390/ijms24054737. Int J Mol Sci. 2023. PMID: 36902165 Free PMC article. Review.
-
Clinical aspects of left ventricular diastolic function assessed by Doppler echocardiography following acute myocardial infarction.Dan Med Bull. 2001 Nov;48(4):199-210. Dan Med Bull. 2001. PMID: 11767125 Review.
Cited by
-
Harnessing mRNA technology for ischemic heart disease: a review of regenerative and protective therapies.Cardiol Plus. 2025 Apr-Jun;10(2):117-128. doi: 10.1097/CP9.0000000000000118. Epub 2025 Jun 30. Cardiol Plus. 2025. PMID: 40599555 Free PMC article. Review.
-
Two promising approaches in the treatment of myocardial infarction: stem cells and gene therapy.Front Cardiovasc Med. 2025 Feb 19;12:1540066. doi: 10.3389/fcvm.2025.1540066. eCollection 2025. Front Cardiovasc Med. 2025. PMID: 40046966 Free PMC article. Review.
-
Direct fibroblast reprogramming: an emerging strategy for treating organic fibrosis.J Transl Med. 2025 Feb 27;23(1):240. doi: 10.1186/s12967-024-06060-3. J Transl Med. 2025. PMID: 40016790 Free PMC article. Review.
References
-
- Mechanic O.J., Gavin M., Grossman S.A. Acute Myocardial Infarction. StatPearls Publishing LLC.; Treasure Island, FL, USA: 2023. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

