Hepatocellular Carcinoma: Beyond the Border of Advanced Stage Therapy
- PMID: 38893154
- PMCID: PMC11171154
- DOI: 10.3390/cancers16112034
Hepatocellular Carcinoma: Beyond the Border of Advanced Stage Therapy
Abstract
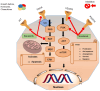
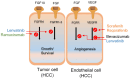
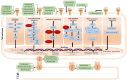
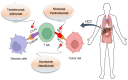
Hepatocellular carcinoma (HCC) is the deadliest emergent health issue around the globe. The stronger oncogenic effect, proteins, and weakened immune response are precisely linked with a significant prospect of developing HCC. Several conventional systemic therapies, antiangiogenic therapy, and immunotherapy techniques have significantly improved the outcomes for early-, intermediate-, and advanced-stage HCC patients, giving new hope for effective HCC management and prolonged survival rates. Innovative therapeutic approaches beyond conventional treatments have altered the landscape of managing HCC, particularly focusing on targeted therapies and immunotherapies. The advancement in HCC treatment suggested by the Food and Drug Administration is multidimensional treatment options, including multikinase inhibitors (sorafenib, lenvatinib, regorafenib, ramucirumab, and cabozantinib) and immune checkpoint inhibitors (atezolizumab, pembrolizumab, durvalumab, tremelimumab, ipilimumab, and nivolumab), in monotherapy and in combination therapy to increase life expectancy of HCC patients. This review highlights the efficacy of multikinase inhibitors and immune checkpoint inhibitors in monotherapy and combination therapy through the analysis of phase II, and III clinical trials, targeting the key molecular pathways involved in cellular signaling and immune response for the prospective treatment of advanced and unresectable HCC and discusses the upcoming combinations of immune checkpoint inhibitors-tyrosine kinase inhibitors and immune checkpoint inhibitors-vascular endothelial growth factor inhibitors. Finally, the hidden challenges with pharmacological therapy for HCC, feasible solutions for the future, and implications of possible presumptions to develop drugs for HCC treatment are reported.
Keywords: combination therapy; hepatocellular carcinoma; hepatocytes; immune checkpoint inhibitors; immune microenvironment; immune system; multikinase inhibitors.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Advances in Immune Checkpoint Inhibitors for Advanced Hepatocellular Carcinoma.Front Immunol. 2022 Jun 10;13:896752. doi: 10.3389/fimmu.2022.896752. eCollection 2022. Front Immunol. 2022. PMID: 35757756 Free PMC article. Review.
-
Immunotherapy and chimeric antigen receptor T-cell therapy in hepatocellular carcinoma.Chin Clin Oncol. 2021 Feb;10(1):11. doi: 10.21037/cco-20-231. Chin Clin Oncol. 2021. PMID: 33541088 Review.
-
Role of immunotherapy in the management of hepatocellular carcinoma: current standards and future directions.Curr Oncol. 2020 Nov;27(Suppl 3):S152-S164. doi: 10.3747/co.27.7315. Epub 2020 Nov 1. Curr Oncol. 2020. PMID: 33343209 Free PMC article. Review.
-
Drug Treatment for Advanced Hepatocellular Carcinoma: First-Line and Beyond.Curr Oncol. 2022 Aug 4;29(8):5489-5507. doi: 10.3390/curroncol29080434. Curr Oncol. 2022. PMID: 36005172 Free PMC article. Review.
-
Immunotherapies for hepatocellular carcinoma.Nat Rev Clin Oncol. 2022 Mar;19(3):151-172. doi: 10.1038/s41571-021-00573-2. Epub 2021 Nov 11. Nat Rev Clin Oncol. 2022. PMID: 34764464 Review.
Cited by
-
Hepatocellular Carcinoma and Antibody Drug Conjugates: A Systematic Review.Cureus. 2025 Apr 24;17(4):e82912. doi: 10.7759/cureus.82912. eCollection 2025 Apr. Cureus. 2025. PMID: 40416124 Free PMC article. Review.
-
Navigating liver cancer: Precision targeting for enhanced treatment outcomes.Drug Deliv Transl Res. 2025 Jun;15(6):1935-1961. doi: 10.1007/s13346-024-01780-x. Epub 2025 Jan 23. Drug Deliv Transl Res. 2025. PMID: 39847205 Review.
-
Effects of sertraline and sorafenib on HepG2 cells with a possible link to autophagy.Med Int (Lond). 2025 May 29;5(4):47. doi: 10.3892/mi.2025.246. eCollection 2025 Jul-Aug. Med Int (Lond). 2025. PMID: 40502851 Free PMC article.
-
Efficacy and safety of regorafenib as a first-line agent alone or in combination with an immune checkpoint inhibitor for advanced hepatocellular carcinoma: a retrospective cohort study.J Gastrointest Oncol. 2024 Jun 30;15(3):1072-1081. doi: 10.21037/jgo-24-315. Epub 2024 Jun 27. J Gastrointest Oncol. 2024. PMID: 38989425 Free PMC article.
-
Immune evasion from macrophages by NEAT1-induced CD24 in liver cancer.Oncogene. 2025 Aug 12. doi: 10.1038/s41388-025-03537-3. Online ahead of print. Oncogene. 2025. PMID: 40796954
References
-
- El-Serag H.B., Balakrishnan M., Natarajan Y. Epidemiology and Risk Factors of Hepatocellular Carcinoma. J. Gastrointest. Oncol. 2024:250–263. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

