Effective Synthesis of High-Integrity mRNA Using In Vitro Transcription
- PMID: 38893337
- PMCID: PMC11173937
- DOI: 10.3390/molecules29112461
Effective Synthesis of High-Integrity mRNA Using In Vitro Transcription
Abstract
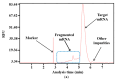
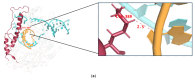
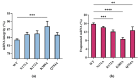
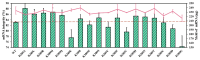
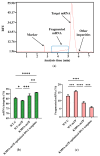
mRNA vaccines are entering a period of rapid development. However, their synthesis is still plagued by challenges related to mRNA impurities and fragments (incomplete mRNA). Most impurities of mRNA products transcribed in vitro are mRNA fragments. Only full-length mRNA transcripts containing both a 5'-cap and a 3'-poly(A) structure are viable for in vivo expression. Therefore, RNA fragments are the primary product-related impurities that significantly hinder mRNA efficacy and must be effectively controlled; these species are believed to originate from either mRNA hydrolysis or premature transcriptional termination. In the manufacturing of commercial mRNA vaccines, T7 RNA polymerase-catalyzed in vitro transcription (IVT) synthesis is a well-established method for synthesizing long RNA transcripts. This study identified a pivotal domain on the T7 RNA polymerase that is associated with erroneous mRNA release. By leveraging the advantageous properties of a T7 RNA polymerase mutant and precisely optimized IVT process parameters, we successfully achieved an mRNA integrity exceeding 91%, thereby further unlocking the immense potential of mRNA therapeutics.
Keywords: T7 RNA polymerase; fragmented mRNA; mRNA integrity; mutation; synthesis; transcription.
Conflict of interest statement
Wei He, Xinya Zhang, Yangxiaoyu Zou, Ji Li, Chong Wang and Qiuheng Jin were employed by Vazyme Biotech Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

