Design, Synthesis, Antifungal Activity, and 3D-QSAR Study of Novel Quinoxaline-2-Oxyacetate Hydrazide
- PMID: 38893377
- PMCID: PMC11173898
- DOI: 10.3390/molecules29112501
Design, Synthesis, Antifungal Activity, and 3D-QSAR Study of Novel Quinoxaline-2-Oxyacetate Hydrazide
Abstract
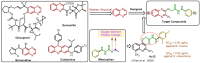
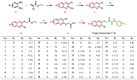
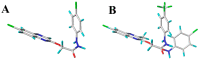
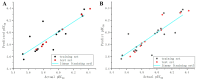
Plant pathogenic fungi pose a major threat to global food security, ecosystem services, and human livelihoods. Effective and broad-spectrum fungicides are needed to combat these pathogens. In this study, a novel antifungal 2-oxyacetate hydrazide quinoxaline scaffold as a simple analogue was designed and synthesized. Their antifungal activities were evaluated against Botrytis cinerea (B. cinerea), Altemaria solani (A. solani), Gibberella zeae (G. zeae), Rhizoctonia solani (R. solani), Colletotrichum orbiculare (C. orbiculare), and Alternaria alternata (A. alternata). These results demonstrated that most compounds exhibited remarkable inhibitory activities and possessed better efficacy than ridylbacterin, such as compound 15 (EC50 = 0.87 μg/mL against G. zeae, EC50 = 1.01 μg/mL against C. orbiculare) and compound 1 (EC50 = 1.54 μg/mL against A. alternata, EC50 = 0.20 μg/mL against R. solani). The 3D-QSAR analysis of quinoxaline-2-oxyacetate hydrazide derivatives has provided new insights into the design and optimization of novel antifungal drug molecules based on quinoxaline.
Keywords: 2-oxyacetate hydrazide; 3D-QSAR; antifungal activity; quinoxaline.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Wu Y.-L., Han L.-J., Wu X.-M., Jiang W., Liao H., Xu Z., Pan C.-P. Trends and perspectives on general Pesticide analytical chemistry. Adv. Agrochem. 2022;1:113–124. doi: 10.1016/j.aac.2022.11.002. - DOI
-
- Wang X., Fu X., Chen M., Wang A., Yan J., Mei Y., Wang M., Yang C. Novel 1,3,5-thiadiazine-2-thione derivatives containing a hydrazide moiety: Design, synthesis and bioactive evaluation against phytopathogenic fungi in vitro and in vivo. Chin. Chem. Lett. 2019;30:1419–1422. doi: 10.1016/j.cclet.2019.03.038. - DOI
-
- Swain T. Secondary Compounds as Protective Agents. Annu. Rev. Plant Biol. 1977;28:479–501. doi: 10.1146/annurev.pp.28.060177.002403. - DOI
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources

