Ag-CeO2 Based on Electrochemical Sensor for High-Efficient On-Site Detection of Nitrite in Aquaculture Water and Beverages
- PMID: 38893519
- PMCID: PMC11173993
- DOI: 10.3390/molecules29112644
Ag-CeO2 Based on Electrochemical Sensor for High-Efficient On-Site Detection of Nitrite in Aquaculture Water and Beverages
Abstract
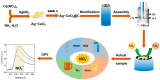
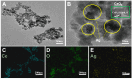
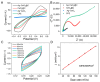
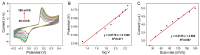
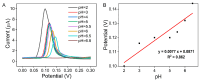
Nitrite is one of the most common nitrogenous compounds, which is not only an important indicator of aquaculture water but also widely used as a food additive. Its potential toxicity poses a huge threat to aquatic products and human health. Therefore, it is important to develop a convenient and rapid sensor for the high-efficient onsite detection of nitrite. In this work, a novel electrochemical sensor was developed for the qualitative and quantitative analysis of nitrite. The developed nitrite electrochemical detection system is easily applied in onsite detection. The electrochemical working electrode was constructed based on the combination of Ag-CeO2 and conductive carbon paste (CPE) with excellent electrocatalysis activity and rapid electron transfer ability. By the application of the developed system and under the optimal conditions, the linear range was from 40.0 μM to 500.0 μM, and the detection limit was reduced to 4.3 μM. The recovery was between 92.1% and 108.1%, and the relative standard deviations (RSDs) were 0.49%~9.31%. The sensor exhibited superior reproducibility, high stability sensitivity, and anti-interference ability, confirming its effectiveness for nitrite analysis. Finally, the developed electrochemical sensor was successfully applied to detect nitrite in beverages and aquaculture water samples, indicating that this approach has great potential in onsite food testing and environmental monitoring.
Keywords: Ag-CeO2; aquaculture water; beverage testing; electrochemical sensor; nitrite.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
A high-performance electrochemical sensor based on dendritic Au/Zn modified carbon cloth for the determination of nitrite in aquaculture.Sci Total Environ. 2024 Nov 10;950:175346. doi: 10.1016/j.scitotenv.2024.175346. Epub 2024 Aug 6. Sci Total Environ. 2024. PMID: 39117201
-
Cerium vanadate/functionalized carbon nanofiber composite for the electrochemical detection of nitrite.Food Chem. 2024 Nov 30;459:140353. doi: 10.1016/j.foodchem.2024.140353. Epub 2024 Jul 17. Food Chem. 2024. PMID: 39024884
-
A novel electrochemical sensor based on silver/halloysite nanotube/molybdenum disulfide nanocomposite for efficient nitrite sensing.Biosens Bioelectron. 2018 Jun 30;109:279-285. doi: 10.1016/j.bios.2018.02.057. Epub 2018 Mar 6. Biosens Bioelectron. 2018. PMID: 29573727
-
Application of Graphene-Based Materials for Detection of Nitrate and Nitrite in Water-A Review.Sensors (Basel). 2019 Dec 20;20(1):54. doi: 10.3390/s20010054. Sensors (Basel). 2019. PMID: 31861855 Free PMC article. Review.
-
Gold Nanomaterials and their Composites as Electrochemical Sensing Platforms for Nitrite Detection.Chem Asian J. 2022 Oct 17;17(20):e202200823. doi: 10.1002/asia.202200823. Epub 2022 Sep 19. Chem Asian J. 2022. PMID: 36039466 Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

