Preparation, Characterization, and Antioxidant Properties of Self-Assembled Nanomicelles of Curcumin-Loaded Amphiphilic Modified Chitosan
- PMID: 38893567
- PMCID: PMC11173681
- DOI: 10.3390/molecules29112693
Preparation, Characterization, and Antioxidant Properties of Self-Assembled Nanomicelles of Curcumin-Loaded Amphiphilic Modified Chitosan
Abstract
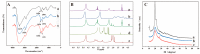
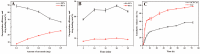
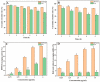
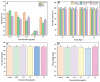
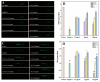
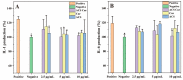
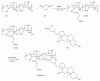
Curcumin (Cur) is a phytochemical with various beneficial properties, including antioxidant, anti-inflammatory, and anticancer activities. However, its hydrophobicity, poor bioavailability, and stability limit its application in many biological approaches. In this study, a novel amphiphilic chitosan wall material was synthesized. The process was carried out via grafting chitosan with succinic anhydride (SA) as a hydrophilic group and deoxycholic acid (DA) as a hydrophobic group; 1H-NMR, FTIR, and XRD were employed to characterize the amphiphilic chitosan (CS-SA-DA). Using a low-cost, inorganic solvent-based procedure, CS-SA-DA was self-assembled to load Cur nanomicelles. This amphiphilic polymer formed self-assembled micelles with a core-shell structure and a critical micelle concentration (CMC) of 0.093 mg·mL-1. Cur-loaded nanomicelles were prepared by self-assembly and characterized by the Nano Particle Size Potential Analyzer and transmission electron microscopy (TEM). The mean particle size of the spherical Cur-loaded micelles was 770 nm. The drug entrapment efficiency and loading capacities were up to 80.80 ± 0.99% and 19.02 ± 0.46%, respectively. The in vitro release profiles of curcumin from micelles showed a constant release of the active drug molecule. Cytotoxicity studies and toxicity tests for zebrafish exhibited the comparable efficacy and safety of this delivery system. Moreover, the results showed that the entrapment of curcumin in micelles improves its stability, antioxidant, and anti-inflammatory activity.
Keywords: amphiphilic chitosan; curcumin; nanomicelles.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence in the work reported in this paper.
Figures

References
-
- Ashrafizadeh M., Zarrabi A., Hashemi F., Moghadam E.R., Hashemi F., Entezari M., Hushmandi K., Mohammadinejad R., Najafi M. Curcumin in cancer therapy: A novel adjunct for combination chemotherapy with paclitaxel and alleviation of its adverse effects. Life Sci. 2020;256:117984. doi: 10.1016/j.lfs.2020.117984. - DOI - PubMed
MeSH terms
Substances
Grants and funding
- 2023A21623/Science and technology development special fund competitive allocation project of Zhanjiang
- 4SG22308P/Songshan Lake Medical and Industrial Integration Innovation project
- GDPKLAPPS2206/Guangdong Provincial Key Laboratory of Aquatic Product Processing and Safety
- 2020ZDB15/Guangdong Provincial Drug Administration key laboratory project
LinkOut - more resources
Full Text Sources
Medical

