CXCR4 Expression as a Prognostic Biomarker in Soft Tissue Sarcomas
- PMID: 38893721
- PMCID: PMC11172351
- DOI: 10.3390/diagnostics14111195
CXCR4 Expression as a Prognostic Biomarker in Soft Tissue Sarcomas
Abstract
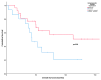
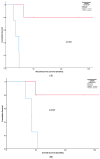
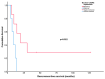
Poor long-term survival in localized high-risk soft tissue sarcomas (STSs) of the extremities and trunk highlights the need to identify new prognostic factors. CXCR4 is a chemokine receptor involved in tumor progression, angiogenesis, and metastasis. The aim of this study was to evaluate the association between CXCR4 expression in tumor tissue and survival in STSs patients treated with neoadjuvant therapy. CXCR4 expression was retrospectively determined by immunohistochemical analysis in serial specimens including initial biopsies, tumors post-neoadjuvant treatment, and tumors after relapse. We found that a positive cytoplasmatic expression of CXCR4 in tumors after neoadjuvant treatment was a predictor of poor recurrence-free survival (RFS) (p = 0.003) and overall survival (p = 0.019) in synovial sarcomas. We also found that positive nuclear CXCR4 expression in the initial biopsies was associated with poor RFS (p = 0.022) in undifferentiated pleomorphic sarcomas. In conclusion, our study adds to the evidence that CXCR4 expression in tumor tissue is a promising prognostic factor for STSs.
Keywords: CXCR4; prognostic factor; soft tissue sarcomas; synovial sarcomas; undifferentiated pleomorphic sarcomas.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Histopathological re-classification of extremity pleomorphic soft tissue sarcoma has clinical relevance.Eur J Surg Oncol. 2004 Dec;30(10):1131-6. doi: 10.1016/j.ejso.2004.07.018. Eur J Surg Oncol. 2004. PMID: 15522563
-
Clinicopathological and prognostic significance of chemokine receptor CXCR4 in patients with bone and soft tissue sarcoma: a meta-analysis.Clin Exp Med. 2017 Feb;17(1):59-69. doi: 10.1007/s10238-015-0405-y. Epub 2015 Dec 17. Clin Exp Med. 2017. PMID: 26678086
-
MRP1 overexpression determines poor prognosis in prospectively treated patients with localized high-risk soft tissue sarcoma of limbs and trunk wall: an ISG/GEIS study.Mol Cancer Ther. 2014 Jan;13(1):249-59. doi: 10.1158/1535-7163.MCT-13-0406. Epub 2013 Oct 21. Mol Cancer Ther. 2014. PMID: 24145283
-
Adjuvant and neoadjuvant chemotherapy for soft tissue sarcomas: JCOG Bone and Soft Tissue Tumor Study Group.Jpn J Clin Oncol. 2021 Feb 8;51(2):180-184. doi: 10.1093/jjco/hyaa231. Jpn J Clin Oncol. 2021. PMID: 33313851 Review.
-
Neoadjuvant chemotherapy in soft tissue sarcomas: latest evidence and clinical implications.Ther Adv Med Oncol. 2017 Jun;9(6):415-429. doi: 10.1177/1758834017705588. Epub 2017 Apr 16. Ther Adv Med Oncol. 2017. PMID: 28607580 Free PMC article. Review.
References
-
- The WHO Classification of Tumours Editorial Board . Soft Tissue and Bone Tumours WHO Classification of Tumours. 5th ed. IARC; Lyon, France: 2020.
-
- Gronchi A., Miah A.B., Dei Tos A.P., Abecassis N., Bajpai J., Bauer S., Biagini R., Bielack S., Blay J.Y., Bolle S., et al. Soft Tissue and Visceral Sarcomas: ESMO–EURACAN–GENTURIS Clinical Practice Guidelines for Diagnosis, Treatment and Follow-Up☆. Ann. Oncol. 2021;32:1348–1365. doi: 10.1016/j.annonc.2021.07.006. - DOI - PubMed
-
- Casali P.G., Abecassis N., Bauer S., Biagini R., Bielack S., Bonvalot S., Boukovinas I., Bovee J.V.M.G., Brodowicz T., Broto J.M., et al. Soft Tissue and Visceral Sarcomas: ESMO-EURACAN Clinical Practice Guidelines for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2018;29:iv51–iv67. doi: 10.1093/annonc/mdy096. - DOI - PubMed
LinkOut - more resources
Full Text Sources

