Chiral Me-2-arachidonoyl Glycerols: The First Potent Endocannabinoid Glyceride Templates with Stability to COX-2
- PMID: 38894922
- PMCID: PMC11181503
- DOI: 10.1021/acsmedchemlett.4c00175
Chiral Me-2-arachidonoyl Glycerols: The First Potent Endocannabinoid Glyceride Templates with Stability to COX-2
Abstract
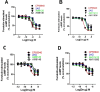
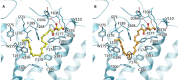
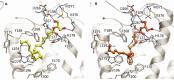
2-Arachidonoyl glycerol (2-AG) is the principal endogenously produced ligand for the cannabinoid CB1 and CB2 receptors (CBRs). The lack of potent and efficacious 2-AG ligands with resistance against metabolizing enzymes represents a significant void in the armamentarium of research tools available for studying eCB system molecular constituents and their function. Herein we report the first endocannabinoid glyceride templates with remarkably high potency and efficacy at CBRs. Two of our lead chiral 2-AG analogs, namely, (13S)- and (13R)-Me-2-AGs, potently inhibit excitatory neurotransmission via CB1 while they are endowed with excellent resistance to the oxidizing enzyme COX-2. Our SAR results are supported by docking studies of the key analog and 2-AG on the crystal structures of CB1.
© 2024 American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
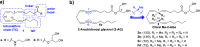


References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

