Identification of novel biomarkers for anti- Toxoplasma gondii IgM detection and the potential application in rapid diagnostic fluorescent tests
- PMID: 38894968
- PMCID: PMC11184589
- DOI: 10.3389/fmicb.2024.1385582
Identification of novel biomarkers for anti- Toxoplasma gondii IgM detection and the potential application in rapid diagnostic fluorescent tests
Abstract
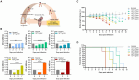
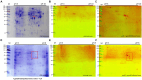
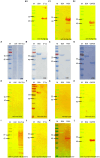
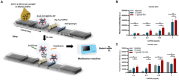
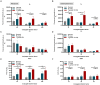
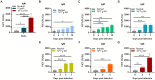
Toxoplasmosis, while often asymptomatic and prevalent as a foodborne disease, poses a considerable mortality risk for immunocompromised individuals during pregnancy. Point-of-care serological tests that detect specific IgG and IgM in patient sera are critical for disease management under limited resources. Despite many efforts to replace the T. gondii total lysate antigens (TLAs) by recombinant antigens (rAgs) in commercial kits, while IgG detection provides significant specificity and sensitivity, IgM detection remains comparatively low in sensitivity. In this study, we attempted to identify novel antigens targeting IgM in early infection, thereby establishing an IgM on-site detection kit. Using two-dimensional gel electrophoresis (2DE) and mouse serum immunoblotting, three novel antigens, including EF1γ, PGKI, and GAP50, were indicated to target T. gondii IgM. However, rAg EF1γ was undetectable by IgM of mice sera in Western blotting verification experiments, and ELISA coated with PGKI did not eliminate cross-reactivity, in contrast to GAP50. Subsequently, the lateral flow reaction employing a strip coated with 0.3 mg/mL purified rAg GAP50 and exhibited remarkable sensitivity compared with the conventional ELISA based on tachyzoite TLA, which successfully identified IgM in mouse sera infected with tachyzoites, ranging from 103 to 104 at 5 dpi and 104 at 7 dpi, respectively. Furthermore, by using standard T. gondii-infected human sera from WHO, the limit of detection (LOD) for the rapid fluorescence immunochromatographic test (FICT) using GAP50 was observed at 0.65 IU (international unit). These findings underline the particular immunoreactivity of GAP50, suggesting its potential as a specific biomarker for increasing the sensitivity of the FICT in IgM detection.
Keywords: 2DE immunoblotting; IgM detection; Toxoplasma gondii; diagnosis; fluorescence immunochromatographic test; point-of-care test.
Copyright © 2024 Nguyen, Yeo and Park.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Alvarado-Esquivel C., Niewiadomski A., Schweickert B., Liesenfeld O. (2011). Antiparasitic treatment suppresses production and avidity of toxoplasma gondii-specific antibodies in a murine model of acute infection*. Eur. J. Microbiol. Immunol. 1, 249–255. doi: 10.1556/EuJMI.1.2011.3.9, PMID: - DOI - PMC - PubMed
-
- Anand N., Lutshumba J., Whitlow M., Abdelaziz M. H., Mani R., Suzuki Y. (2022). Deficiency in indoleamine-2, 3-dioxygenase induces upregulation of guanylate binding protein 1 and inducible nitric oxide synthase expression in the brain during cerebral infection with toxoplasma gondii in genetically resistant BALB/c mice but not in genetically susceptible C57BL/6 mice. Microbes Infect. 24:104908. doi: 10.1016/j.micinf.2021.104908, PMID: - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

